US Pharm. 2017;42(10):42-46.
ABSTRACT: The management of type 2 diabetes (T2DM) has evolved significantly over the past several decades. One of the newest additions to antidiabetic therapy is a sodium-glucose cotransporter 2 (SGLT2) inhibitor with a unique mechanism that targets the kidney’s ability to reabsorb filtered glucose. In addition to providing glycemic control, this class has a unique mechanism of action associated with blood pressure reduction, weight loss, and potential cardiovascular benefits; however, the FDA is closely monitoring the use of these drugs based on increased safety concerns. The benefits and risks of SGLT2 inhibitors should be carefully considered. Selected patients with T2DM can benefit from SGLT2 inhibitor therapy.
Diabetes, a devastating disease affecting more than 29 million Americans, is a growing epidemic and the leading cause of end-stage renal disease, lower-limb amputation, and blindness in the United States. In addition, cardiovascular (CV) events are highly prevalent in patients with diabetes.1
Metformin is the recommended first-line drug for type 2 diabetes (T2DM) patients with poor glycemic control. However, the glycemic response produced by metformin is typically inadequate in the long-term management of diabetes, and patients will eventually require additional treatments. The need for newer agents to treat T2DM is now being recognized. Most agents used for T2DM either improve the body’s sensitivity to insulin or increase pancreatic secretion of insulin. Improved understanding of the mechanisms involved in glucose reabsorption through the kidneys has led to the development of sodium-glucose cotransporter 2 (SGLT2) inhibitors. At this writing, three agents in this class have been approved by the FDA: canagliflozin, dapagliflozin, and empagliflozin (TABLE 1).2,3

Mechanism of Action
More than 99% of glucose entering the kidneys and reaching the glomeruli is filtered through the nephrons. In healthy patients, the proximal tubules reabsorb glucose from the filtered urine before reaching the loop of Henle. Glycosuria occurs when a surge in plasma glucose leads to an increase in the filtered glucose reaching the kidneys, overwhelming the proximal tubules’ resorptive capacity. Until recently, the mechanism of glucose reabsorption was not fully understood but was thought to be achieved by coupling sodium transport across the membrane with glucose.3 It is now clear that the reabsorption of glucose in the proximal tubules into renal tubular epithelial cells is managed by the adenosine triphosphate–dependent protein family (SGLTs).1 In contrast to high-affinity and low-capacity SGLT1, low-affinity and high-capacity SGLT2 is responsible for most of the glucose reabsorption in the renal tubules. In the last few years, several molecules were developed to specifically target SGLT2s for the treatment of T2DM.3 SGLT2 inhibitors decrease the reabsorption of glucose and lower the renal threshold for glucose, consequently escalating urinary glucose excretion.4
Glycemic Efficacy
According to the American Diabetes Association (ADA), SGLT2 inhibitors have intermediate efficacy in lowering glucose.5 Reducing blood glucose (BG) concentrations through the glycosuric effect is independent of insulin secretion or action. SGLT2 inhibitors were investigated in a number of randomized, controlled trials (RCTs) both as monotherapy and in combination with metformin, sulfonylureas, pioglitazone, sitagliptin, and insulin.
Compared with placebo, SGLT2 inhibitors significantly reduced A1C by approximately 0.6% to 1.0% and also reduced fasting plasma glucose (FPG) concentrations at the end of the 24- and 26-week studies.6-8 Even when 1- and 2-year results were evaluated, SGLT2 inhibitors had favorable glycemic effects on A1C and FPG concentrations.2 SGLT2 inhibitors were also compared with other antidiabetic agents (sulfonylureas and sitagliptin). Head-to-head trials of SGLT2 inhibitors versus sulfonylureas and sitagliptin showed similar-to-favorable glycemic control when A1C and FPG reductions were assessed in patients with T2DM.8-11 Glycemic benefits persisted in 1- and 2-year evaluations.2
A number of studies also examined the efficacy of SGLT2 inhibitors as add-on therapy. Adding an SGLT2 inhibitor to an antidiabetic regimen involving metformin, sulfonylureas, pioglitazone, sitagliptin, or insulin led to a greater A1C reduction without a significant increase in hypoglycemia risk.12-16
The glycemic efficacy of SGLT2 inhibitors is clearly shown in many RCTs. The benefits of SGLT2 inhibitors in patients with T2DM are not limited to their glucose-lowering effect, but also include reductions in blood pressure (BP) and body weight (discussed next).
Reductions in BP and Weight
Several RCTs of SGLT2 inhibitors have evaluated changes in systolic BP (SBP) and weight as secondary endpoints. In RCTs that compared SGLT2 inhibitors with placebo, SGLT2 inhibitors significantly reduced SBP by approximately 3 to 5 mmHg and weight by roughly 4 to 6 pounds.5-7 Positive effects on weight and BP also were seen in RCTs comparing SGLT2 inhibitors with other antidiabetic agents.8
The BP-lowering effect is most likely due to osmotic diuresis, which is believed to contribute to the initial weight loss as well. Gradual weight loss is thought to be caused by a reduction in adipose tissue as well as calorie loss through glycosuria.8 These additional benefits of SGLT2 inhibitors are theorized to contribute to positive CV outcomes; accordingly, a number of trials are investigating the CV benefit of SGLT2 inhibitors.
SGLT2 Inhibitors and CV Outcomes
One study that evaluated the CV benefit of SGLT2 inhibitors was the Empagliflozin Cardiovascular Outcome Event Trial in Type 2 Diabetes Mellitus Patients.17 This RCT examined the effect of empagliflozin on CV morbidity and mortality in older patients with long-standing T2DM and established CV disease. Approximately 7,000 adults were randomized to one of three groups (empagliflozin 10 mg daily, empagliflozin 25 mg daily, or placebo) and followed for a median of 3.1 years. The primary outcome was a composite of deaths from CV causes, nonfatal myocardial infarction (MI) excluding silent MI, and nonfatal stroke.17
In this RCT, the pooled empagliflozin group had significantly lower mortality rates than the placebo group (14% reduction in composite death). Also, a significant reduction in death from CV causes was observed in the pooled empagliflozin group (3.7% vs. 5.9% in the placebo group, translating to a 38% relative risk reduction) that was primarily driven by lower rates of heart failure–related hospitalization or death.17 In December 2016, as a result of empagliflozin’s potential CV benefits, the FDA approved an expanded indication for empagliflozin to reduce the risk of CV death in adult patients with T2DM and established CV disease.
More recently, another cardiovascular-outcome trial of SGLT2 inhibitors, the Canagliflozin Cardiovascular Assessment Study, was completed. This RCT included more than 10,000 patients with T2DM and high CV risk who were randomized to either canagliflozin or placebo and followed for a mean of 3.6 years. The rate of the primary outcome—a composite of death from CV causes, nonfatal MI, or nonfatal stroke—was reduced by 14%, and it was concluded that canagliflozin may confer a renal benefit. However, there was an almost twofold increase in the risk of amputation (mainly at the level of the toe or metatarsal) with canagliflozin use.18
These two trials have shown CV benefits from SGLT2 inhibitors. It is, however, premature to conclude that this CV benefit is a drug-class effect of SGLT2 inhibitors; future CV-outcome studies of SGLT2 inhibitors will help determine this.
Safety Concerns
The most common adverse reactions associated with SGLT2 inhibitors are polyuria and genitourinary infections. A high concentration of glucose in filtered urine causes osmotic diuresis, which leads to polyuria and contributes to volume depletion. It is important to recognize that uncontrolled T2DM patients may present with polyuria.4,19,20
Postmarketing data include reports of serious urinary tract infections (UTIs) with SGLT2 inhibitors, including urosepsis and pyelonephritis requiring hospitalization. Consequently, the FDA revised SGLT2 inhibitor labeling to warn about the risk of serious UTIs. In addition, patients taking SGLT2 inhibitors, especially females and uncircumcised males, are at increased risk for genital mycotic infections, likely because of the high glucose concentration in excreted urine. Signs and symptoms of UTIs and genital infections should be evaluated and treated promptly.3,4,19,20
SGLT2 inhibitors cause intravascular volume depletion. As a result, symptomatic hypotension can occur, especially in patients with reduced renal function (TABLE 2), elderly patients, patients taking diuretics or medications that interfere with the renin-angiotensin-aldosterone system (RAAS), and patients with low SBP. Volume status should be assessed prior to and during SGLT2 inhibitor therapy.4,19,20
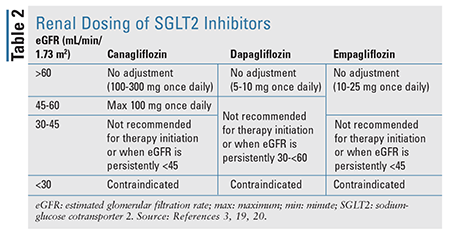
Volume depletion also may lead to renal impairment. Acute kidney injury (AKI) requiring hospitalization and dialysis has been reported in patients taking SGLT2 inhibitors. Based on these reports, the FDA has strengthened the existing warning about the risk of AKI with SGLT2 inhibitor use. In addition, concomitant use of medications that interfere with potassium excretion or the RAAS can put patients at risk for developing hyperkalemia, especially in the case of concurrent renal impairment. Patients with hypovolemia, chronic renal insufficiency or congestive heart failure or who are taking concomitant nephrotoxic medications should not be started on SGLT2 inhibitors. Renal function should be evaluated before SGLT2 inhibitors are initiated and periodically during use.4,15,19,20
Reports of ketoacidosis, including fatal cases, have surfaced in postmarketing surveillance of SGLT2 inhibitors. Unlike typical diabetic ketoacidosis, SGLT2 inhibitor–associated ketoacidosis presents with euglycemia or only modestly elevated BG concentrations (often <250 mg/dL), which causes delayed detection and treatment of ketoacidosis. Clinicians should keep this in mind and discontinue SGLT2 inhibitors immediately if ketoacidosis is suspected. It may be necessary to temporarily withhold SGLT2 inhibitors in clinical situations—such as prolonged fasting due to acute illness or surgery—that are known to predispose patients to ketoacidosis.4,19,20
SGLT2 inhibitors increase the risk of hypoglycemia when they are used concurrently with insulin and insulin secretagogues. To minimize the risk of hypoglycemia, the doses of insulin and insulin secretagogue should be reduced upon initiation of SGLT2 inhibitors.4,19,20
Postmarketing reports on canagliflozin prompted the FDA to strengthen the warning for increased risk of bone fractures.21 However, a new meta-analysis including all three SGLT2 inhibitors found that the increased risk of bone fractures associated with SGLT2 inhibitors was not significant. Long-term RCTs and real-world data are warranted for more definitive conclusions.22
The FDA recently issued a new warning that canagliflozin increases the risk of leg and foot amputations. Patients should be instructed to notify their providers of any new pain or tenderness, sores or ulcers, or infections in the legs or feet. Healthcare professionals should evaluate predisposing factors such as history of prior amputation, peripheral vascular disease, neuropathy, and diabetic foot ulcers.23
Place in Therapy
Both the 2015 update to the ADA/European Association for the Study of Diabetes (EASD) position statement and the 2017 ADA diabetes care guidelines have classified SGLT2 inhibitors as second-line agents after metformin.5,24 The ADA guidelines also state that empagliflozin should specifically be considered for patients with T2DM and established atherosclerotic CV disease to reduce CV and all-cause mortality (level of evidence B), but they include a cautionary warning about the uncertainty of a drug-class effect related to CV protection.5
The American Association of Clinical Endocrinologists (AACE) and the American College of Endocrinology (ACE) also provided a consensus statement on the comprehensive T2DM-management algorithm, which discusses the efficacy and safety of SGLT2 inhibitors and their place in therapy. Unlike the ADA/EASD statement and the ADA guidelines, the 2017 AACE/ACE consensus statement suggests a hierarchy of antidiabetic drug usage. Of all available antidiabetic drugs, including oral and injectable agents, AACE/ACE placed SGLT2 inhibitors as third-line treatment after metformin and glucagon-like peptide-1 receptor agonists. Therefore, per the AACE/ACE guidelines, SGLT2 inhibitors are preferred to alternative antidiabetic drugs.25
In addition to the recommendations of national guidelines, providers should carefully assess the additional benefits of SGLT2 inhibitors versus safety concerns and drug interactions (TABLE 3). Since SGLT2 inhibitors are relatively new and available as brand only, cost should also be considered.5,24,25

Counseling
Patients should be counseled to report symptoms of ketoacidosis, hypotension, UTI, and genital mycotic infections. Clinicians should advise patients to maintain adequate fluid intake because decreased oral intake or increased loss of fluids can potentially lead to AKI. In addition, patients must be informed of common side effects, such as increased urination, increased thirst, nausea, and constipation. Patients taking SGLT2 inhibitors, especially canagliflozin, should be advised to inform their providers of any new pain, sores, ulcers, or infections affecting their legs or feet.4,19,20
Conclusion
SGLT2 inhibitors used in the treatment of T2DM have a unique mechanism that specifically targets the kidneys. The safety profile of this drug class is well documented in the literature and is mostly consistent with these medications’ mechanism of action. The most common adverse effects include genital mycotic infections, UTIs, and effects of intravascular volume depletion. Hypoglycemia, the most common side effect of antidiabetic agents, is fairly uncommon with SGLT2 inhibitors given as monotherapy. Serious safety issues are not commonly reported, although new problems are still being recognized. In addition to glycemic efficacy, empagliflozin and canagliflozin recently demonstrated CV benefits in T2DM patients. The use of SGLT2 inhibitors is becoming more prominent in the treatment of T2DM because of additional benefits such as low hypoglycemia risk, decreased mortality secondary to CV events, and efficacy in lowering BG concentrations, body weight, and BP. However, recent safety warnings raise concerns about the use of SGLT2 inhibitors in T2DM management. Risks and benefits must be closely evaluated in order to appropriately use SGLT2 inhibitors in patients with T2DM.
REFERENCES
1. CDC. Diabetes: working to reverse the US epidemic at a glance 2016. www.cdc.gov/chronicdisease/resources/publications/aag/diabetes.htm. Accessed June 6, 2017.
2. Liu XY, Zhang N, Chen R, et al. Efficacy and safety of sodium-glucose cotransporter 2 inhibitors in type 2 diabetes: a meta-analysis of randomized controlled trials for 1 to 2 years. J Diabetes Complications. 2015;29:1295-1303.
3. Hardman TC, Dubrey SW. Development and potential role of type-2 sodium-glucose transporter inhibitors for management of type 2 diabetes. Diabetes Ther. 2011;2:133-145.
4. Invokana (canagliflozin) package insert. Titusville, NJ: Janssen Pharmaceuticals, Inc; 2013.
5. American Diabetes Association. Pharmacologic approaches to glycemic treatment. Standards of medical care in diabetes—2017. Diabetes Care. 2017;40(suppl 1):S64-S74.
6. Stenlöf K, Cefalu WT, Kim KA, et al. Efficacy and safety of canagliflozin monotherapy in subjects with type 2 diabetes mellitus inadequately controlled with diet and exercise. Diabetes Obes Metab. 2013;15:372-382.
7. Ferrannini E, Ramos SJ, Salsali A, et al. Dapagliflozin monotherapy in type 2 diabetic patients with inadequate glycemic control by diet and exercise: a randomized, double-blind, placebo-controlled, phase 3 trial. Diabetes Care. 2010;33:2217-2224.
8. Roden M, Weng J, Eilbracht J, et al. Empagliflozin monotherapy with sitagliptin as an active comparator in patients with type 2 diabetes: a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Diabetes Endocrinol. 2013;1:208-219.
9. Lavalle-González FJ, Januszewicz A, Davidson J, et al. Efficacy and safety of canagliflozin compared with placebo and sitagliptin in patients with type 2 diabetes on background metformin monotherapy: a randomised trial. Diabetologia. 2013;56:2582-2592.
10. Cefalu WT, Leiter LA, Yoon KH, et al. Efficacy and safety of canagliflozin versus glimepiride in patients with type 2 diabetes inadequately controlled with metformin (CANTATA-SU): 52 week results from a randomised, double-blind, phase 3 non-inferiority trial. Lancet. 2013;382:941-950.
11. Del Prato S, Nauck M, Durán-Garcia S, et al. Long-term glycaemic response and tolerability of dapagliflozin versus a sulphonylurea as add-on therapy to metformin in patients with type 2 diabetes: 4-year data. Diabetes Obes Metab. 2015;17:581-590.
12. Jabbour SA, Hardy E, Sugg J, Parikh S; Study 10 Group. Dapagliflozin is effective as add-on therapy to sitagliptin with or without metformin: a 24-week, multicenter, randomized, double-blind, placebo-controlled study. Diabetes Care. 2014;37:740-750.
13. Bailey CJ, Gross JL, Pieters A, et al. Effect of dapagliflozin in patients with type 2 diabetes who have inadequate glycaemic control with metformin: a randomised, double-blind, placebo-controlled trial. Lancet. 2010;375:2223-2233.
14. Strojek K, Yoon KH, Hruba V, et al. Effect of dapagliflozin in patients with type 2 diabetes who have inadequate glycaemic control with glimepiride: a randomized, 24-week, double-blind, placebo-controlled trial. Diabetes Obes Metab. 2011;13:928-938.
15. Rosenstock J, Vico M, Wei L, et al. Effects of dapagliflozin, an SGLT2 inhibitor, on HbA(1c), body weight, and hypoglycemia risk in patients with type 2 diabetes inadequately controlled on pioglitazone monotherapy. Diabetes Care. 2012;35:1473-1478.
16. Wilding JP, Woo V, Soler NG, et al; Dapagliflozin 006 Study Group. Long-term efficacy of dapagliflozin in patients with type 2 diabetes mellitus receiving high doses of insulin: a randomized trial. Ann Intern Med. 2012;156:405-415.
17. Zinman B, Wanner C, Lachin JM, et al; EMPA-REG OUTCOME Investigators. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. New Engl J Med. 2015;373:2117-2128.
18. Neal B, Perkovic V, Mahaffey KW, et al; CANVAS Program Collaborative Group. Canagliflozin and cardiovascular and renal events in type 2 diabetes. New Engl J Med. 2017; 377:644-657.
19. Farxiga (dapagliflozin) package insert. Wilmington, DE: AstraZeneca Pharmaceuticals LP; August 2016.
20. Jardiance (empagliflozin) package insert. Ridgefield, CT: Boehringer Ingelheim Pharmaceuticals, Inc; December 2016.
21. FDA. FDA Drug Safety Communication: FDA revises label of diabetes drug canagliflozin (Invokana, Invokamet) to include updates on bone fracture risk and new information on decreased bone mineral density. www.fda.gov/Drugs/DrugSafety/ucm461449.htm. Accessed June 3, 2017.
22. Tang HL, Li DD, Zhang JJ, et al. Lack of evidence for a harmful effect of sodium-glucose co-transporter 2 (SGLT2) inhibitors on fracture risk among type 2 diabetes patients: a network and cumulative meta-analysis of randomized controlled trials. Diabetes Obes Metab. 2016;18:1199-1206.
23. FDA. Canagliflozin (Invokana, Invokamet): Drug Safety Communication—increased risk of leg and foot amputations. www.fda.gov/Safety/MedWatch/SafetyInformation/SafetyAlertsforHumanMedicalProducts/ucm558605.htm?source=govdelivery&utm_medium=email&utm_source=govdelivery. Accessed June 5, 2017.
24. Inzucchi SE, Bergenstal RM, Buse JB, et al. Management of hyperglycemia in type 2 diabetes, 2015: a patient-centered approach: update to a position statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care. 2015;38:140-149.
25. Garber AJ, Abrahamson MJ, Barzilay JI, et al. Consensus statement by the American Association of Clinical Endocrinologists and American College of Endocrinology on the comprehensive type 2 diabetes management algorithm—2017 executive summary. Endocr Pract. 2017;23:207-238.
To comment on this article, contact rdavidson@uspharmacist.com.





