US Pharm. 2010;35(5)(Diabetes suppl):14-17.
If not already seeing them, pharmacists should anticipate encountering prescriptions for U-500 regular insulin. U-500 insulin is five times more potent than standard U-100 and is utilized in patients requiring high doses of insulin. Prescribing of U-500 is on the rise due to the obesity epidemic, adoption of tighter glucose control protocols, increased insulin resistance, and utilization of insulin pumps.1 Potential for adverse drug events exists due to unfamiliarity with U-500 and its higher potency. This review is intended to enable safe provision of U-500 insulin by increasing awareness of the drug and its correct administration.
As insulin resistance worsens, greater doses of insulin are required to meet glycemic goals. This is especially true for persons with insulin resistance (IR) syndromes. These are patients with an insulin requirement of 200 U/day and greater. FIGURE 1 illustrates the body’s response to insulin as the dose increases. Note that for patients with IR syndromes, the dose response to insulin is significantly diminished. This is especially true when insulin doses exceed 100 U. However, this phenomenon does not mean greater doses are without effect.2 In fact, higher doses of insulin eventually achieve therapeutic targets. For this reason, use of high-dose insulin should not be avoided in insulin-resistant patients. Otherwise, these patients will fail to achieve glycemic goals by underdosing insulin.
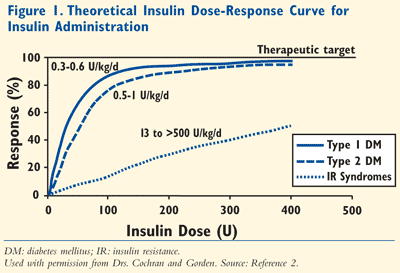
Use of standard insulin becomes problematic when patients require over 200 U/day or greater than 100 U per injection. At these doses, the volume of U-100 insulin is physically too large for single subcutaneous administration, and multiple injections are required to deliver a single dose. With more injections, compliance suffers and poor glycemic control results.3
Fortunately, an alternative exists to treat highly insulin-resistant patients more efficiently. U-500 insulin, manufactured by Eli Lilly, contains 500 U of regular insulin per mL.4 The high concentration enables administration of large doses of insulin in a small volume. U-500 is intended only for subcutaneous administration. IV use is not recommended due to risk of overdose. It is available in 20 mL vials only.4
Pharmacokinetics
U-500 is nonmodified regular insulin. Its pharmacokinetics, however, resemble basal rate delivery similar to NPH insulin.2 The high concentration of U-500 results in delayed absorption and is responsible for a slow rate of delivery.4-6 Single doses of U-500 are generally active for 8 hours, although activity can last up to 24 hours.2-4 The range is due to variability in blood flow at the injection site and sensitivity of insulin receptors.2 Initial effects of U-500 are similar to regular insulin, with an onset of 30 minutes and peak effects seen within 1 to 3 hours.3 Based on its unique pharmacokinetic properties, U-500 is dosed similar to NPH, with 2 to 3 injections per day.2
Clinical Trials
There are no large prospective trials investigating U-500; however, data available from a handful of small studies confirm its safety and efficacy. As is typical in clinical practice, the trials involve switching a small number of insulin-resistant patients from U-100 to U-500. Neal reported an average HbA1C decrease of 1.7 %, with a nonsignificant increase in body mass index (BMI).7 Wafa and Kahn observed a 2.2% decrease in HbA1C, with a small weight gain of 1.6% after 12 months.8 Ballani et al reported an HbA1C decrease of 2.5% along with a significant average weight gain of 4.7 kg.9 On average, switching patients from standard insulin to U-500 resulted in a reduction in A1C of about 2%, with a relatively small increase in weight. Hypoglycemic episodes were either not reported or deemed insignificant compared to standard regimens. The trials confirm improvement in glycemic targets for difficult-to-control patients with diabetes switched from standard insulin to U-500 without significant adverse effect. Increased patient satisfaction related to fewer and lower-volume injections is contributory to the improvements in glycemic control seen with U-500 insulin.3
Pump Patients
U-500 insulin represents an efficient option for insulin pump patients who require high doses of insulin. Highly insulin-resistant patients using standard insulin are inconvenienced by frequent reservoir changing, thereby diminishing the practicality of an insulin pump.3 Due to its high concentration, U-500 permits less frequent filling of pump reservoirs. A few trials demonstrate efficacy and support use of U-500 via continuous subcutaneous infusion.10,11 Clinicians and patients should be aware that insulin pumps are currently only programmed for U-100 insulin. Therefore, one must remember to multiply the units displayed by five when U-500 is loaded in an insulin pump. Also, bolus dosing in a pump is calibrated for rapid-acting insulin, so the clinician must take into account the delayed activity of U-500 when programming.12 Pump patients require follow-up and management by a team familiar with the nuances of U-500 insulin.

Dosing
There are various dosing algorithms for converting from standard insulin to U-500 insulin. One approach is by Garg et al.3 First, add up the total daily dose of U-100 from all sources. Subtract 10% to 20% from this sum to use for initial dosing. Next, divide by 500 to arrive at the number of mL to administer per day. Finally, divide this daily total in 2 to 3 doses. For example, a patient injecting a total of 250 U of U-100 converts to 0.2 mL of U-500 injected twice daily. Follow the example in SIDEBAR 1 and use a dose conversion chart (TABLE 1) to confirm.
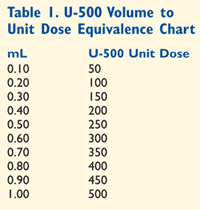
When converting a patient to U-500, all other insulin preparations are discontinued. U-500 becomes the patient’s sole exogenous insulin source. Oral antidiabetic drugs that increase insulin sensitivity should be continued and are especially valuable for obese patients.2 Continue dietary and exercise interventions.
U-500 dose adjustments are not made based on solitary blood glucose readings or carbohydrate counting. Rather, the clinician looks for weekly patterns in glucose readings. For example, if glucose values are consistently below 70 mg/dL in the morning, the evening or predinner dose of U-500 is adjusted downward. Conversely, if glucose readings are consistently high, the preceding insulin dose is adjusted upward.2 The dosing strategy for U-500 is similar to NPH or basal insulin adjustments.
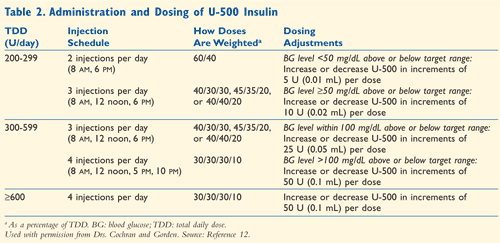
Cochran and Gorden provide an algorithm for the administration and dosing of U-500 insulin (TABLE 2).12 It provides options for division of total daily doses as well as dose recommendations based on migration from glucose targets. Guidelines of pump dosing are available in the original reference, but were not included in this review. The algorithm from Cochran and Gorden is driven by the patient’s total daily dose of insulin and provides a helpful reference to employ when treating patients with U-500 insulin.
Cost
U-500 insulin represents a cost advantage over standard insulin.10 Although it is more expensive per mL, economic analysis concludes that U-500 costs less per unit.12 A reason for this is the reduced volume required per injection compared to U-100. Other cost savings are realized because U-500 is utilized as the sole insulin, compared to standard insulin regimens utilizing basal and bolus insulin. Finally, fewer injections per day translate to decreased cost of syringes and supplies.10
Potential Problems and Recommendations
It is not difficult to imagine the potential for errors or adverse effects when utilizing concentrated U-500 insulin. Fortunately, due to the highly insulin-resistant nature of U-500 patients, hypoglycemia is not a common problem.2 In fact, underdosing and hyperglycemia materialize when patients are admitted to the hospital and inpatient staff are reluctant to prescribe U-500. For users of U-500, however, the potential for hypoglycemic reactions 18 to 24 hours after injection is a concern due to the drug’s delayed kinetics.12 Monitoring for this phenomenon with adjustments to dose and schedule is important.
One area for mishap surrounds the type of syringe utilized. Standard insulin syringes are marked for U-100, so when filled with U-500 insulin, the dose injected does not equal the units printed on the syringe. The doses are, in fact, five times greater. Patients may misinterpret their dose of U-500 insulin as 40 U, for example, when in reality they are injecting 200 U. To avoid ambiguity, express the dose in volume and use tuberculin syringes marked in volume. Tuberculin syringes may not be as readily available as insulin syringes, and some insurers may not regard them as diabetic supplies.12 Regardless, pharmacies should stock 0.5 and 1.0 mL tuberculin syringes with 30-gauge, 0.5-inch needles and provide them to patients using U-500 insulin.
Prescription Labeling
SIDEBAR 2 details an accurately written and labeled prescription for U-500. Note that the provider wrote the actual number of units intended for injection. Additionally, the prescription is labeled to express dose in volume as well as number of units. Unfortunately, prescriptions for U-500 are frequently written less clearly. Consider the same example, but written as: Rx U-500, inject “30 units” SQ tid. In this interpretation, 30 units with quotation marks refers to the unit markings on a U-100 syringe. This is confusing, because the patient is actually injecting 150 U of insulin. Patients and providers familiar with U-500 may understand the distinction between “30 units” and 150 actual units, but it is possible for a caregiver or another clinician to misinterpret the dose. Therefore, the dose of U-500 should be expressed in both volume and total units. Further, add a warning statement to the prescription label, such as “Concentrated insulin—do not adjust dose.” It is up to the pharmacist to label U-500 prescriptions as clearly as possible.

Inappropriate Product Selection and Stocking
The Institute for Safe Medication Practices has reported an increase in the number of mix-ups between U-100 and U-500 insulin.1 Mistakes occur when prescribers or pharmacists mistakenly select U-500 for U-100. Adjacent drug entries in computer databases contribute to inappropriate selection. To correct this, list U-500 separately from other insulin and add obvious characters to focus attention to the concentration. When able, consider electronically turning off U-500 to avoid inappropriate selection during routine order processing.
Within the pharmacy, there are potential pitfalls to stocking U-500. If you do not have patients requiring U-500, do not stock it. Otherwise, store U-500 away from standard insulin and label the storage tray to remind staff of the drug’s unique concentration. Fortunately, the box containing U-500 displays distinctive diagonal orange stripes and is labeled “not for ordinary use.”
Counseling
The most important safety intervention a pharmacist can perform when dispensing U-500 insulin is conveyance of effective patient counseling. This should include family and caregivers. When counseling about U-500, ensure that all parties involved are aware that this insulin is five times more concentrated than U-100.3 Next, demonstrate how to draw up the exact dose using a tuberculin syringe. The patient should reproduce the effort to ensure proficiency. Vials of saline and test syringes are useful for effective demonstration. Carefully teach the correct dose in both volume and units of insulin. Make sure the patient and caregiver know that U-500 doses should not be adjusted without the advice of a physician. Review proper subcutaneous injection technique and supply alcohol prep pads with a sharps container. Finally, remind patients that U-500 insulin vials should be discarded 30 days after opening. No matter how well labeled, misunderstood prescriptions result in medication errors. Thus, effective counseling is of paramount concern. SIDEBAR 3 provides pharmacists with a U-500 dispensing and counseling checklist for review.

Summary
U-500 insulin fits a pharmacotherapy niche for highly insulin-resistant patients. Notably, it solves the volume-related problem associated with high-dose U-100 insulin. Use of U-500 insulin is increasing in relation to a growing diabetic and obesity population. Pharmacists should promote safe administration of U-500 through careful labeling and counseling. Using tuberculin syringes and expressing the dose in volume and units enhances safety. With patient education, use of U-500 need not be feared and in fact is recommended for those patients requiring high doses of insulin to achieve glycemic goals.
REFERENCES
1. Humulin R Concentrate U-500. Medication Safety Alert, Community/Ambulatory Care Edition. Institute for Safe Medication Practices. August 30, 2007. www.ismp.org/Newsletters/
2. Cochran E, Musso C, Gorden P. The use of U-500 in patients with extreme insulin resistance. Diabetes Care. 2005;28:1240-1244.
3. Garg R, Johnston V, McNally PG, et al. U-500 insulin: why, when and how to use in clinical practice. Diabetes Metab Res Rev. 2007;23:265-268.
4. Humulin R insulin (U-500) package insert. Indianapolis, IN: Eli Lilly and Company; 2009.
5. Galloway JA, Spradlin CT, Nelson RL, et al. Factors influencing the absorption, serum insulin concentration, and blood glucose responses after injections of regular insulin and various insulin mixtures. Diabetes Care. 1981;4:366-376.
6. Nathan DM, Axelrod L, Flier JS, et al. U-500 insulin in the treatment of antibody-mediated insulin resistance. Ann Intern Med. 1981;94:653-656.
7. Neal JM. Analysis of effectiveness of human U-500 insulin in patients unresponsive to conventional insulin therapy. Endocr Pract. 2005;11:305-307.
8. Wafa WS, Khan MI. Use of U-500 regular insulin in type 2 diabetes. Diabetes Care. 2006;29:2175-2176.
9. Ballani P, Tran MT, Navar MD, Davidson MB. Clinical experience with U-500 regular insulin in obese, markedly insulin-resistant type 2 diabetic patients. Diabetes Care. 2006;29:2504-2505.
10. Knee TS, Seidensticker DF, Walton JL, et al. A novel use of U-500 insulin for continuous subcutaneous insulin infusion in patients with insulin resistance: a case series. Endocr Pract. 2003;9:181-186.
11. Lane WS. Use of U-500 regular insulin by continuous subcutaneous infusion in patients with type 2 diabetes and severe insulin resistance. Endocr Pract. 2006;12:251-256.
12. Cochran E, Gorden P. Use of U-500 insulin in the treatment of severe insulin resistance. Insulin. 2008;3:211-218.
To comment on this article, contact rdavidson@uspharmacist.com.





