US Pharm. 2016;41(4):18-21.
ABSTRACT: Urinary tract infections (UTIs) are very common in women and may be classified as acute (uncomplicated), recurrent, or complicated. Due to increasing antibiotic resistance, beta-lactam therapy has become less effective. There have also been recent reports of drug-induced UTIs related to sodium-glucose cotransporter 2 (SGLT2) inhibitors. Treatment of complicated UTIs (cUTIs) has become increasingly complex because of the rising prevalence of multidrug-resistant gram-negative bacteria. Two new IV antibiotics (Zerbaxa, Avycaz) have the potential to overcome some of the antibiotic resistance noted with oral medications. Both of these drugs combine an antibiotic with novel beta-lactamase inhibitors and have shown promise in eradicating gram-negative resistant infections.
Urinary tract infections (UTIs) represent a significant health problem in both community and hospital-based settings. Approximately 11% of women in the United States report a minimum of one physician-diagnosed UTI per year, and the lifetime probability of UTI in females is 60%.1
Background
These infections may involve the lower urinary tract, the upper urinary tract, or both. The presence of bacteriuria without symptoms is termed asymptomatic bacteriuria. Cystitis refers to infections limited to the lower urinary tract presenting with dysuria, frequent and urgent urination, and occasional suprapubic tenderness. Acute pyelonephritis refers to infection of the upper urinary tract accompanied by fever, flank pain, and significant bacteriuria. Complicated UTI (cUTI) occurs in patients with predisposing factors including obstructed urinary flow (i.e., congenital causes), prostatic obstruction or urinary stones, incomplete bladder emptying due to anatomical or neurogenic causes, foreign bodies within the urinary tract (e.g., catheters, drainage tubes), systemic illness (e.g., diabetes), and pregnancy.1
Causes and Risk Factors
Urine is normally sterile, but infection may occur due to bacterial entry from the periurethral area. The most common causative organisms include Escherichia coli, Klebsiella pneumoniae, Staphylococcus aureus, Candida species, and Mycobacterium tuberculosis. Risk factors for UTIs include previous UTI, sexual activity (particularly with new sexual partners), use of spermicides, menopause, pregnancy, reduced mobility (i.e., after surgery or bed rest), urinary incontinence, kidney stones, and prostate enlargement. Other risk factors include age (older adults are at higher risk) and gender. Female patients are at higher risk compared to males because of shorter urethra length and proximity of the urethra to the anus, increasing the risk that bacteria will enter the urinary tract.2 Elderly patients may present with atypical symptoms including changes in mental status, lethargy, weakness, and abdominal pain.3,4
A new class of drugs for the treatment of type 2 diabetes has been linked to increased cases of UTIs in patients. Sodium-glucose cotransporter 2 (SGLT2)inhibitors or “flozins” are oral hypoglycemics that work by increasing the amount of glucose spilled into the urine. This class of drugs includes canagliflozin (Invokana), empagliflozin (Jardiance), and dapagliflozin (Farxiga). The FDA recently highlighted this risk in a Drug Safety Communication in May 2015, reminding prescribers to evaluate patients for signs and symptoms of UTI, treat promptly if indicated, and counsel patients on how to recognize the signs and symptoms related to UTIs. Unfortunately, in some patients these UTIs may develop into life-threating renal complications or failure, as well as urosepsis.5
Treatment
Historically, the treatment for uncomplicated acute cystitis has consisted of 7 to 10 days of antibiotic treatment. However, studies have shown that a 3-day course of antimicrobials has equivalent efficacy to longer treatment courses, with eradication rates of >90%.6 Due to increasing antibiotic resistance, beta-lactam therapies such as first-generation cephalosporins and amoxicillin have become less effective than other agents listed in TABLE 1.1
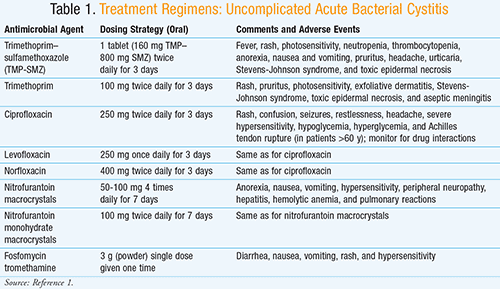
Recurrent UTIs are defined as two or more UTIs within 6 months, or three or more UTIs within a 12-month period. Strategies to prevent recurrent UTIs in women include continuous prophylaxis, postcoital prophylaxis, and acute self-treatment. A continuous prophylaxis strategy consists of using antibiotic treatment once daily, every other night, or 3 nights per week.7 This strategy may reduce recurrent UTIs by 95%, but may be associated with uropathogen resistance.8 The postcoital prophylaxis option consists of a single dose of antibiotic taken within 2 hours of intercourse and may be most beneficial for women who experience UTIs within 24 to 48 hours after intercourse. Reliable patients who have experienced well-documented, recurrent infection and are in good communication with their healthcare provider may benefit from an acute self-treatment approach with initiation of treatment when symptoms occur. In these situations, patients receive a prescription for a 3-day dosage regimen and start therapy when symptoms develop.1
Complicated UTIs and Antimicrobial Resistance
Treatment of cUTIs has become increasingly complex due to the rising prevalence of multidrug-resistant (MDR) gram-negative bacteria. The CDC has identified specific serious public health threats including extended spectrum beta-lactamases (ESBLs) and the organism Pseudomonas aeruginosa, and has labeled carbapenemase-resistant Enterobacteriaceae (CRE) an urgent threat.9
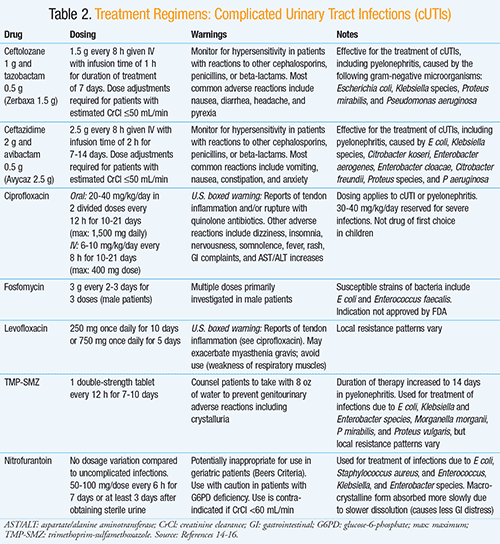
Clinicians are advised to consider local resistance patterns during selection of antimicrobial therapy (TABLE 2), particularly for E coli since this microbe is responsible for 75% to 95% of uncomplicated cystitis and pyelonephritis. However, treatment of uncomplicated UTIs based upon data from hospital antibiograms remains an “imperfect science.” For example, these data may be skewed due to inclusion of samples from inpatient populations or patients with complicated infections. In addition, local in vitro resistance rates may change over time or be unknown. Thus, additional options for optimal care include identification of predictors of resistance unique to an individual patient, such as use of trimethoprim-sulfamethoxazole (TMP-SMZ) in the preceding 3 to 6 months or travel outside the U.S.10
Prevention
Methods to prevent UTIs include urination before and after sexual activity, hydration, regular urination, taking showers instead of baths, and avoidance of douching or use of sprays and/or powders in the genital area. In addition, females should be taught to wipe from front to back during toilet training activities.2
A recent study published in 2015 highlighted the role of diet in the prevention (or treatment strategy) of UTIs.11 This study identified two factors important in a patient’s ability to resist bacterial growth. First, urine with higher pH (i.e., increased levels of alkalinity) was shown to be of importance. The pH of urine may be increased by intake of certain drugs (such as calcium supplements), and alkalinizing agents are available as OTC options in other countries such as the United Kingdom. A second finding in the study involved the protein siderocalin, which is produced by humans during UTIs and blocks bacterial iron uptake. Foods rich in polyphenols such as tea, coffee, wine, and cranberries affect siderocalin’s antibacterial activity and may be helpful as a strategy to prevent UTIs or treat these infections without antibiotics.11
Patients who wish to utilize alternative treatments such as cranberry juice may approach pharmacists for advice. In these situations, counseling points include selection of products with reduced sugar and calories, such as a “light” juice or cocktail. Cranberry tablets or capsules may provide an alternative for patients who wish to avoid cranberry juice.
New IV Antibiotics
In December 2014, the FDA approved a new drug to treat cUTIs. Zerbaxa (TABLE 2) combines an antibiotic to eradicate bacteria (ceftolozane) and a beta-lactamase inhibitor (tazobactam) to combat the enzymes produced by bacteria to resist the antibiotic. A recent study (ASPECT–cUTI phase III trial) compared Zerbaxa to levofloxacin and found that Zerbaxa was superior to levofloxacin in achieving the primary outcome of clinical cure and microbiologic eradication. This combination was also highly effective against multiresistant and levofloxacin-resistant pathogens. Specifically, only 2.7% of gram-negative pathogens were resistant to Zerbaxa, compared to 26.7% resistant to levofloxacin. Common adverse effects include nausea, diarrhea, headache, and fever.12
The FDA also approved another fixed-combination antimicrobial agent, ceftazidime-avibactam (Avycaz), with multiple indications including cUTIs (TABLE 2). This is the fifth antimicrobial agent approved under the Generating Antibiotic Incentives Now (GAIN) title of the FDA Safety and Innovation Act.13 The inclusion of avibactam restores the ability of ceftazidime, a third-generation cephalosporin, to show activity against numerous beta-lactamase producing gram-positive bacteria.9 Avycaz is effective in treating cUTIs caused by gram-negative microorganisms including E coli, Klebsiella species, and P aeruginosa. As part of an effort to reduce further emergence of drug-resistant bacteria and to retain the effectiveness of these new IV antibiotics, pharmacists should advise prescribers that both Zerbaxa and Avycaz be reserved for patients with infections proven (or strongly suspected) to be caused by susceptible bacteria.14,15
Conclusion
The release of two new IV antibiotics with activity against MDR bacteria is a welcome addition to the pharmacologic treatment of UTIs. Pharmacists must maintain vigilance when counseling patients about antibiotic adherence and drug-induced causes of UTI, as well as increasing their efforts to reserve these new antibiotics for infections caused by designated susceptible microorganisms.
REFERENCES
1. Treatment of urinary tract infections in nonpregnant women. ACOG Practice Bulletin. Obstet Gynecol. 2008;111(3):785-794.
2. CDC. Get smart: know when antibiotics work. Urinary tract infection. www.cdc.gov/getsmart/community/for-patients/common-illnesses/uti.html. Accessed January 31, 2016.
3. Matthews SJ, Lancaster JW. Urinary tract infections in the elderly population. Am J Geriatr Pharmacother. 2011;9:286-309.
4. Nicolle LE. Urinary tract infections in the elderly. Clin Geriatr Med. 2009;25:423-436.
5. FDA Drug Safety Communications. FDA revises labels of SGLT2 inhibitors for diabetes to include warnings about too much acid in the blood and serious urinary tract infections. December 4, 2015. www.fda.gov/Drugs/DrugSafety/ucm475463.htm. Accessed January 31, 2016.
6. Iravani A, Tice AD, McCarty J, et al. Short-course ciprofloxacin treatment of acute uncomplicated urinary tract infection in women. The minimum effective dose. Arch Intern Med. 1995;155(5):485-494.
7. Epp A, Larochelle A, Lovatsis D, et al. Recurrent urinary tract infection. J Obstet Gynaecol Can. 2010;32:1082-1090.
8. Lichtenberger P, Hooton TM. Antimicrobial prophylaxis in women with recurrent urinary tract infections. Int J Antimicrob Agents. 2011;38(suppl):36-41.
9. Mawal Y, Critchley IA, Riccobene TA, et al. Ceftazidime–avibactam for the treatment of complicated urinary tract infections and complicated intra-abdominal infections. Expert Rev Clin Pharm. 2015;8(6):691-707.
10. Gupta K, Hooton TM, Naber KG, et al. International clinical practice guidelines for the treatment of acute uncomplicated cystitis and pyelonephritis in women: a 2010 update by the Infectious Diseases Society of America and the European Society for Microbiology and Infectious Disease. Clin Infect Dis. 2011;52(5):e103-e120.
11. Shields-Cutler RR, Crowley JR, Hung CS, et al. Human urinary composition controls antibacterial activity of siderocalin. J Biol Chem. 2015;290(26):15949-15960.
12. Edwards JK. Ceftolozane-tazobactam gives a new ASPECT to the fight against antimicrobial resistance. Nat Rev Nephrol. 2015;11:388.
13. FDA approves new antibacterial drug Zerbaxa. FDA news release. December 19, 2014. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm427534.htm. Accessed January 31, 2016.
14. Avycaz (ceftazidime and avibactam) package insert. Cincinnati, OH: Forest Pharmaceuticals, Inc; September 2015.
15. Zerbaxa (ceftolozane and tazobactam) package insert. Whitehouse Station, NJ: Steri-Pharma, LLC; July 2015.
16. Lexi-Drugs [subscription database]. Hudson, OH: Lexi-Comp, Inc; 2016. http://online.lexi.com. Accessed January 31, 2016.
To comment on this article, contact rdavidson@uspharmacist.com.





