US Pharm. 2017;42(10):8-10.
Currently, public health is at risk on a global scale owing to the presence of counterfeit medications in the medical-product supply chain (MPSC). According to the World Health Organization (WHO), the scope of this problem is enormous: substandard, spurious, falsely labeled, falsified, and counterfeit medical products are difficult to detect because they are frequently designed to appear identical to the genuine product and may not cause an identifiable adverse reaction; however, they often fail to effectively treat the disease or condition for which they were intended.1
In the United States, the FDA promotes and protects the public health by ensuring that safe, effective, and high-quality products are available to the American public, including vulnerable older adults.2 However, protecting the integrity of the MPSC is very complex, requiring a global approach in the current pharmaceutical marketplace.3 Depending on the region of the world, the trafficking of counterfeit medicines is reported to be either a crime of opportunism or a global criminal enterprise of organized and complex proportions.4,5
An ongoing, ever-heightened vigilance is necessary to secure the MPSC since a significant percentage of FDA-approved prescription-drug products are manufactured outside of the U.S. Pharmacists have an important role to play in this mission to keep poor-quality and counterfeit medications out of circulation by reporting adverse events related to the use of suspect medicines and alerting their patients and customers to report suspect products obtained from questionable sources (see Resource 1). Pharmacy-school educators, including preceptors, can involve students in their introductory and advanced pharmacy-practice experiences in efforts and projects to raise public awareness and guide older adults in appropriate safety measures. A checklist for patients and their caregivers with safety tips from the WHO regarding medications purchased through the Internet is available in Resource 2.

Counterfeit Drugs
Drug affordability, particularly for older adults, is often cited as the reason drugs are sought and purchased through the Internet. The FDA has stated that it believes a better job can be done to make safe and innovative drugs more affordable in the U.S. for older adults and other consumers; however, there is a balance that must be achieved such that solutions do not jeopardize safety in an effort to achieve increased affordability.6
When patients purchase medications through the Internet, it significantly compounds the problem of battling the presence of counterfeit medications in the MPSC.6 This is not a new problem. Testimony before Congress in 2003 regarding the FDA’s efforts to assess and respond to the public-health threats noted that the FDA, during a drug-importation survey, found that a majority of parcels at several mail facilities contained unapproved drugs with the potential to cause considerable safety hazards.
According to the testimony of John M. Taylor, III, then Associate Commissioner for Regulatory Affairs at the FDA, “Drugs previously withdrawn from the U.S. market for safety reasons; animal drugs sold illegally for human use; drugs improperly packaged in sandwich bags or tissue paper; drugs without English labeling or proper instructions for use; and drugs requiring precise dosing and monitoring by a physician” were found.6
Using 2009–2011 data from the Counterfeiting Incident System (CIS) database of the Pharmaceutical Security Institute, researchers assessed counterfeit reports involving the legitimate supply chain.7 They identified 1,510 CIS reports involving counterfeits, with 27.6% reporting China as the source country of the incident/detection.7 Most reports originated from health-related government agencies. The most prevalent therapeutic category was anti-infectives (21.1%), followed by genitourinary (14.5%), cardiovascular (11.6%), and central nervous system (11.0%) categories; all therapeutic categories were represented.7
According to Mackey and colleagues, officials were not surprised at the high frequency with which the anti-infective category occurred in these reports, in light of private- and public-sector observations that lifesaving drugs—those products that treat or cure diseases generally associated with potential morbidity or mortality, including both branded and generic anti-infectives/antibiotics—are often reported as counterfeit.1,8-11 To review a table of the legitimate supply-chain counterfeit penetration by therapeutic category, and to view a global map of CIS counterfeit incidents by country, refer to Table 1 and Figure 1, respectively, in Reference 7.
The Drug Supply Chain Security Act
The Drug Supply Chain Security Act (DSCSA) addresses requirements for manufacturers, repackagers, wholesale distributors, dispensers, and third-party logistics providers (trading partners).12,13 While some DSCSA requirements commenced in 2014 and 2015, the remaining requirements, the development of standards, and the system for product tracing will continue to be phased in until 2023. The FDA will continue working with trading partners and other stakeholders on effective implementation.
Some examples of the major provisions of the DSCSA regarding tracing are:
• Product tracing (by 2015, lot-level; by 2023, package-level) and enhanced systems (electronic, interoperable systems to trace products at the package level by 2023)
• Verification requirements change once a product is serialized (starting in 2017 for manufacturers; 2018 for repackagers; 2019 for wholesale distributors; and 2020 for dispensers)
For further information on the DSCSA, refer to References 12 and 13.
FDA Supply Chain Security Toolkit:
Protecting Medical-Product Integrity
Supply-chain problems need to be combated so that the public, including older adults, can have confidence in the integrity of prescription drugs that are vital for health and well-being. The charge of keeping poor-quality and counterfeit medications out of the supply chain falls on the compliance office of the FDA Center for Drug Evaluation and Research. To that end, the FDA led the design and development of the Supply Chain Security Toolkit to be used by industry stakeholders and regulators worldwide to encourage adoption of best practices; for training purposes; and to strengthen laws and regulations to protect consumers from unsafe and substandard drug products.3,14
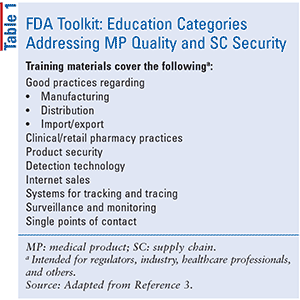
Further training for and use of the toolkit will be rolled out with the establishment of Asia Pacific Economic Cooperation Training Centers of Excellence for Regulatory Science (CoEs); education and training categories are outlined in TABLE 1 and additional information about CoEs is available within the toolkit.3
Conclusion
Keeping the U.S. prescription-drug supply chain among the safest in the world is an enormously challenging responsibility. The importation of counterfeit drugs—manufactured and distributed by well-financed and well-organized networks—is increasing. Foreign Internet sites become portals for unsafe and illegal drugs. This public-health risk is a considerable challenge for the FDA. Pharmacists are key players and need to do their part in this massive collaborative effort in trying to prevent counterfeit medications from potentially harming older adults and the public at large.
REFERENCES
1. World Health Organization. Substandard, spurious, falsely labeled, falsified and counterfeit (SSFFC) medical products. Fact sheet. Updated January 2016. www.who.int/mediacentre/factsheets/fs275/en/. Accessed September 5, 2017.
2. FDA. Health care provider alert: another counterfeit cancer medicine found in United States. Purchasing unapproved drugs is risky business. Updated September 25, 2013. www.fda.gov/Drugs/ResourcesForYou/Consumers/BuyingUsingMedicineSafely/CounterfeitMedicine/ucm338283.htm.Accessed July 25, 2017.
3. FDA. FDA leads effort to create a supply chain security toolkit for medical products. Updated July 20, 2017. www.fda.gov/drugs/drugsafety/drugintegrityandsupplychainsecurity/ucm559053.htm. Accessed July 25, 2017.
4. United Nations Office of Drugs and Crime. Transnational organized crime in East Asia and the Pacific: a threat assessment. 2013. www.unodc.org/toc/en/reports/TOCTA-EA-Pacific.html. Accessed September 4, 2017.
5. Interpol. Trafficking in illicit goods and counterfeiting. www.interpol.int/Crime-areas/Trafficking-in-illicit-goods-and-counterfeiting/Trafficking-in-illicit-goods-and-counterfeiting. Accessed September 4, 2017.
6. Drug importation. Statement of John M. Taylor, III, Associate Commissioner for Regulatory Affairs, FDA, before the Senate Committee on Commerce, Science and Transportation. November 20, 2003. Updated: June 29, 2009. https://wayback.archive-it.org/7993/20170723044410/https://www.fda.gov/NewsEvents/Testimony/ucm114937.htm. Accessed September 13, 2017.
7. Mackey TK, Liang BA, York P, et al. Counterfeit drug penetration into global legitimate medicine supply chains: a global assessment. Am J Trop Med Hyg. 2015;92(suppl 6):59-67.
8. Mackey TK. Global health diplomacy and the governance of counterfeit medicines: a mapping exercise of institutional approaches. J Health Diplomacy. 2013;1:1-20.
9. Newton PN, Green MD, Fernández FM. Impact of poor-quality medicines in the “developing” world. Trends Pharmacol Sci. 2010;31:99-101.
10. Nayyar GM, Breman JG, Newton PN, Herrington J. Poor-quality antimalarial drugs in southeast Asia and sub-Saharan Africa. Lancet Infect Dis. 2012;12:488-496.
11. Dorlo TPC, Ravinetto RM, Beijnen JH, Boelaert M. Substandard medicines are the priority for neglected tropical diseases. BMJ. 2012;345:e7518.
12. Are you ready for the Drug Supply Chain Security Act? Updated: June 30, 2017.
www.fda.gov/Drugs/DrugSafety/DrugIntegrityandSupplyChainSecurity/DrugSupplyChainSecurityAct/ucm427033.htm. Accessed September 4, 2017.
13. Drug Supply Chain Security Act (Title II of the Drug Quality and Security Act) Overview of product tracing requirements. September 2015.
www.fda.gov/downloads/drugs/drugsafety/drugintegrityandsupplychainsecurity/drugsupplychainsecurityact/UCM464907.htm. Accessed September 4, 2017.
14. Bernstein I. Keeping the U.S. prescription drug supply chain among the safest in the world. Posted July 20, 2017. FDA Voice. https://blogs.fda.gov/fdavoice/index.php/2017/07/. Accessed September 4, 2017.
To comment on this article, contact rdavidson@uspharmacist.com.







