Inflammatory bowel disease (IBD) is a condition that causes chronic inflammation in the lining of the gastrointestinal tract (GIT). IBD encompasses two distinct yet very similar diseases: ulcerative colitis and Crohn's disease.1,2 Ulcerative colitis primarily involves the rectum, lower colon, or the entire large intestine and is characterized by diffuse inflammation to the superficial mucosal layers.1 In comparison, Crohn's disease involves any area of the GIT from the oral cavity to the anus but is limited primarily to the colon with or without small intestine disease. Additionally, the inflammation in Crohn's disease is often described as transmural , inflicting damage through each mucosal layer of the GIT.2 Therapeutic intervention for IBD includes the use of pharmacological agents and/or surgery. However, in patients with Crohn's disease, surgery is reserved for those who have medically refractory disease.
Epidemiology
The incidence and prevalence of Crohn's disease is highest in the United Kingdom, northern Europe, and North America. The incidence of Crohn's disease in North America ranges from 3.1 to 14.6 cases per 100,000 person-years, which translates to approximately 10,000 to 47,000 new diagnoses per year. With a prevalence of 29 to 199 cases per 100,000 persons, it is estimated that more than 630,000 people in the United States and Canada have Crohn's disease. 3
It has been theorized that underdeveloped countries have a lower incidence and prevalence of Crohn's disease than do industrialized nations. For example, African countries have an incidence of 0.5 to three cases per 100,000 person-years, and Asian countries have a prevalence ranging from 3.6 to 50.6 cases per 100,000 person-years.3 Crohn's disease is mostly diagnosed during late adolescence and early adulthood and is most pronounced during the second to fourth decade of life.3,4 However, Crohn's disease has been diagnosed in people 50 or older. It has been estimated that the incidence in those older than 60 is 2.5 cases per 100,000 person-years. 5 This disease often affects Caucasians and African-Americans more often than Asians or Hispanics.3,6 There is an equal gender distribution for those diagnosed with Crohn's disease, yet in some epidemiological studies, a slightly higher trend may be observed for women.5,6
Etiology
The pathogenesis of Crohn's disease is unknown; however, environmental, immunological, and genetic factors have been implicated. Smokers are twice as likely to develop symptoms of Crohn's disease compared with nonsmokers.3,4,7 Consumption of certain foods containing high amounts of lactose or fructose may also be a risk factor. In addition, countries that have adopted a more "Westernized diet" (e.g., fast foods, junk foods) are experiencing an increase in the incidence of Crohn's disease.8 Medications, specifically oral contraceptives and measles vaccines, may increase the risk of Crohn's disease. 3 An abnormal or weak immune system may explain the pathogenesis of Crohn's disease, as it has been observed that the intestinal lining of patients with active disease is infiltrated with lymphocytes and mast cells. Other factors that indicate an immunological basis include frequent complaints of extraintestinal conditions, such as arthritis, in patients with Crohn's disease, and the effectiveness of immunosuppressive therapy in the treatment of IBD. Genetic factors may play a role, as there is a high rate of concordance observed in monozygotic twins compared with dizygotic twins. Also, first-degree relatives of those with IBD are more likely to be affected than those who do not have a family history of IBD.9
Clinical Presentation
The signs and symptoms of Crohn's disease--i.e., abdominal cramping, abdominal pain, anorexia, cachexia, diarrhea, epigastric pain, fatigue, fever and other flu-like symptoms, joint pain, pallor, nausea with or without vomiting, rectal bleeding, weight loss--may manifest as mild, moderate, or severe, depending on the location and extent of the disease. The Crohn's Disease Activity Index may be used to determine the severity of the disease in adults. This survey evaluates eight specific signs and symptoms (e.g., frequency of stools passed per week, hematocrit levels, antidiarrheal use, weight); however, because it is time-consuming and complex in the clinical setting, it is mostly used for study purposes. Patients often seek medical attention when they experience abdominal pain and frequent bowel movements with or without rectal bleeding. Patients may also experience ocular (e.g., episcleritis, uveitis), dermatologic (e.g., erythema nodosum, pyoderma gangrenosum), hepatic (e.g., sclerosing cholangitis), and rheumatological (e.g., ankylosing spondylitis, peripheral arthritis, sacroiliitis) extraintestinal complications. Other rare, but not uncommon, complications include toxic megacolon, perianal fistulas, intestinal obstruction, and hypercoagulability. Clinical manifestations caused by malabsorption of nutrients may promote osteomalacia, osteoporosis, hypocalcemia, and pernicious anemia.
Diagnosis
People with complaints of diarrhea, abdominal pain, and rectal bleedings, or other symptoms of IBD should undergo an evaluation. The diagnosis of Crohn's disease is based on patient's symptoms as well as radiographic, endoscopic, pathological, and laboratory findings. Radiographic studies are used to determine the location of the disease and if masses, abscesses, or obstruction are present. However, to confirm the diagnosis, a colonoscopy should be performed to evaluate for cobblestone-like, erythematous, and friabile appearance of the colonic mucosa. A lower endoscopy is also useful in assessing the location of the disease; however, viewing areas from the anus to the terminal ileum are limited. If disease in the small bowel is suspected, a capsule endoscopy may be utilized. This noninvasive procedure--approved by the FDA in August 2001--requires the patient to swallow a disposable miniature video camera contained in a capsule about the size of a large vitamin. As the capsule naturally passes through the GIT, images of the small intestines are captured and recorded digitally. Since many patients with Crohn's disease have active infections or anemia, laboratory monitoring is required.
Disease Management
Although there is no cure for Crohn's disease, there are several goals of treatment. The primary goal is to induce and maintain remission by resolving acute inflammatory processes. Other goals of treatment include improving quality of life by minimizing symptoms of the disease, treating and preventing extraintestinal disorders, and correcting nutritional deficiencies (e.g., malabsorption of calcium, folate, vitamin B12, iron, and zinc).
Several treatment options are available for Crohn's disease; these include corticosteroids, anti-inflammatory agents (e.g., sulfasalazine, mesalamine), immunomodulators, antibiotics, and biological agents (see Table 1). Determining which therapy to initiate depends on the location of the disease, as many of the agents target specific parts of the GIT. In addition, the severity of the disease, (mild to moderate, moderate to severe, or severe to fulminant) is also a factor used to determine which treatment to prescribe.10 A treatment algorithm that guides the clinician to determine which agent to use based on the severity of disease is depicted in Figure 1.
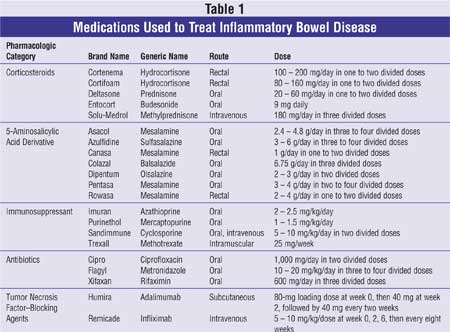
Corticosteroids: Corticosteroids are considered the main treatment for inducing remission and stopping disease progression in individuals having moderate to severe Crohn's disease activity (Figure 1). This class of agents, including prednisone, prednisolone, and methylprednisolone, is commonly used for its immediate onset of action in reducing intestinal inflammation. Remission may be observed at four to eight weeks in approximately 75% to 95% of patients when receiving either an oral or parenteral corticosteroid regimen that is equivalent to 40 to 60 mg per day of prednisone (Table 1).11,12 Although there is a high response rate associated with steroids, this therapy is not recommended for maintaining remission due to its adverse effect profile and its relative ineffectiveness when compared with other agents.10,13 The short-term side effects associated with corticosteroids include acne, adrenal axis suppression, edema, increased appetite, weight gain, hypertension, and mood changes. Approximately 40% to 50% of patients may be steroid dependent.10 These individuals are more likely to develop cataracts, diabetes, muscle atrophy, and bone demineralization (osteopenia and osteoporosis). Young patients at risk for bone loss should receive at least 1,000 mg of elemental calcium daily, and those older than 50 should receive 1,500 mg of elemental calcium supplementation in three divided doses with 400 to 800 IU of vitamin D daily.14 Bisphosphonates, including alendronate, risedronate, and etidronate, should be recommended for those who have received more than three months of steroid therapy or cannot discontinue steroid therapy.14 Alternatively, budesonide may be considered. This synthetic corticosteroid has a high first-pass hepatic metabolism effect resulting in a lower systemic bioavailability, thus causing fewer adverse effects. Budesonide is most effective for Crohn's disease confined to the ileum and ascending colon.15,16 Topical formulations, including suppositories, foams, or enemas, may also be considered to minimize the side effects of corticosteroids.
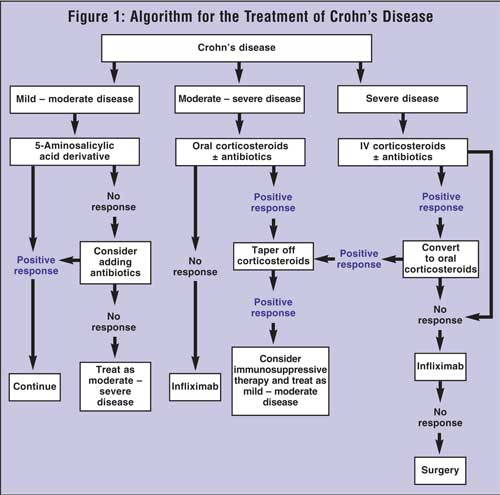
Aminosalicylates: Aminosalicylates are prescribed to maintain remission in patients with mild to moderate IBD (Figure 1). These agents are indicated for ulcerative colitis and are commonly used off label for Crohn's disease. Sulfasalazine was the first aminosalicylate used for IBD and was coincidently found to have anti-inflammatory properties in the GIT when used to treat rheumatoid arthritis. This agent is composed of sulfapyridine and 5-aminosalicylic acid (5-ASA or mesalamine) linked by an azo-bond. Once sulfasalazine reaches the colon, colonic bacterial azo-reductase cleaves the azo-bond separating the inactive (sulfapyridine) and active (mesalamine) parts of the compound.17 Sulfapyridine accounts for most of the dose-related adverse events, which include headaches, nausea, vomiting, anorexia, and dyspepsia. To prevent the untoward effects, initiate lower doses and increase the dose over a period of time to the desired amount. Additionally, administering sulfasalazine with meals may alleviate side effects. Rarely, hypersensitivity reactions (fever, rash, hepatotoxicity, bone marrow suppression, changes in sperm morphology) that are not dose-related may occur. Individuals allergic to sulfonamides should avoid sulfasalazine. Additionally, patients lacking the enzyme glucose-6-phosphate dehydrogenase (G6PD) should avoid sulfasalazine, because hemolytic anemia may develop. Also, sulfasalazine inhibits folate absorption; thus, folate supplementation is recommended to prevent deficiency and anemia.
A number of mesalamine preparations that are sulfonamide-free have since been developed that have a more tolerable side effect profile. All of the agents contain mesalamine but are different in terms of their sites of action in the GIT (Table 2). There may be a role for aminosalicylates in treating mildly active Crohn's disease. In a landmark trial, 43% of the patients receiving high-dose Pentasa at 4 g/day demonstrated a clinical remission compared with only 18% in the placebo arm.18 However, sulfasalazine and mesalamine, when compared with placebo, have not been found to be effective in preventing disease flare-ups after corticosteroid-induced remission.10,17,19,20
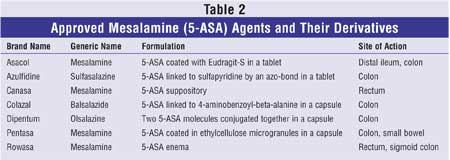
Immunomodulators: Immunosuppressant therapies, including azathioprine and its metabolite 6-mercapto purine (6-MP) are effective in maintaining remission in patients with Crohn's disease (Figure 1).10,21,22 These agents have also demonstrated a steroid-sparing effect in patients who are steroid dependent.23,24 However, it may take up to three to six months before any benefit is observed. The doses used for IBD are typically less than those used for immunosuppression posttransplantation. For Crohn's disease, the most effective dose of azathioprine ranges from 2 to 2.5 mg/kg/day, and that for 6-MP ranges from 1 to 1.5 mg/kg/day.10 The adverse effects associated with the immunosuppressants are rare but may potentially be life-threatening, including drug-induced hepatotoxicity, hypersensitivity reactions, bone marrow suppression, infection, and pancreatitis. The events are reversible with treatment discontinuation. Thus, to prevent these untoward events, many clinicians start with a low dose and escalate to the desired dose with frequent laboratory monitoring. At present, no head-to-head trials have been published to determine if this method of initiating treatment causes less toxicity than starting at the calculated dose.10,24 More recently, thiopurine methyltransferase (TPMT) and 6-methylmercaptopurine (6-MMP), enzymes that metabolizes 6-MP and azathioprine, have been implicated for their clinical response and adverse effects.25 The likelihood of developing adverse events is dependent on an individual's genetic polymorphism for metabolizing these enzymes. Therefore, genetic testing for TPMT and 6-MMP enzymes may be useful in preventing bone marrow and hepatic toxicity as well as detecting compliance. The genetic tests should not replace frequent laboratory monitoring, especially complete blood counts and liver function tests.
Approximately 25% to 50% of patients who do not tolerate either azathioprine or 6-MP therapy benefit from methotrexate.26 Intramuscular methotrexate, 25 mg per week, was found to induce remission in 70% of the patients who were resistant to azathioprine and corticosteroid-dependent.27 Subcutaneous or oral routes may be used; however, these routes are associated with variable therapeutic responses. The most common side effects of methotrexate include nausea and abnormal liver function tests. Hepatotoxicity is a well-documented adverse effect associated with long-term use of methotrexate. Bone marrow suppression is also a known effect that occurs with treatment beyond one year. Since methotrexate is a folic acid antagonist, folate supplementation is recommended.24
Cyclosporine doses greater than 5 mg/kg/day were found to be effective in inducing remission in patients with active Crohn's disease;28 however, relapse occurs frequently upon discontinuing therapy.29 One advantage cyclosporine has over azathioprine, 6-MP, and methotrexate is its quick onset of action. A response is usually observed within two to four weeks of initiating therapy. 30 However, this medication has several adverse effects, including nephrotoxicity, paresthesias, and hypertension. Thus, this treatment should be reserved for use when all other agents for treating Crohn's disease have failed.

Antibiotics: The most commonly used antibiotics for IBD are metronidazole and ciprofloxacin (Figure 1). Their use for active Crohn's disease remains controversial, as most of the published trials are uncontrolled with small sample sizes. The majority of trials that evaluated metronidazole compared with placebo showed no significant difference in achieving a remission response.31 Doses used in the trials ranged from 10 to 20 mg/kg/day, with higher doses showing more efficacy.32 Nausea, metallic taste, and disulfuram-like reaction are the most common side effects of metronidazole but are reversible upon discontinuing therapy. Peripheral neuropathy is a rare condition associated with metronidazole that may not cease with treatment discontinuation.
Ciprofloxacin, a quinolone antibiotic with gram-negative coverage and immunomodulatory effects, has been commonly used in Crohn's disease. However, it has not been documented to be effective in inducing remission.31 In a retrospective study, subjects receiving a combination of metronidazole and ciprofloxacin had a 70.6% clinical response, similar to the 69% response seen with ciprofloxacin only.33
Rifaximin, a rifamycin-derived antibiotic, has undergone several small trials evaluating its effects in patients with IBD.34 In an open-label study evaluating rifaximin 200 mg administered three times daily for 16 weeks, clinical remission at the end of treatment was observed in 59% of the 29 patients with active disease. 35 The use of antibiotics remains to be elucidated, and more controlled trials are needed to determine their usefulness for Crohn's disease.
Tumor Necrosis FactorBlocking Agents: Infliximab was the first of its class to be approved in the U.S. for the treatment of moderate to severe Crohn's disease. This biological agent is a chimeric monoclonal antibody that binds to tumor necrosis factor alpha, thereby neutralizing its inflammatory effect in the gastrointestinal mucosa. In the first randomized placebo-controlled trial, 81% of patients receiving a 5-mg/kg infusion of infliximab had a clinical response four weeks after initiating the first dose.36 Unfortunately, after a single infusion of infliximab, the clinical response is not sustained.37 It has been suggested that the response would be better maintained with repeated infliximab infusions. This was confirmed by the ACCENT I (A Crohn's disease Clinical trial Evaluating infliximab in a New long-term Treatment regimen) trial.38 The results demonstrated that subjects who received infliximab infusions of either 5 or 10 mg/kg every eight weeks were more likely to sustain a clinical remission, 39% and 45% respectively, compared with 19% in the placebo group.38 Additionally, subjects who received placebo were more likely to lose the clinical response sooner than those who received maintenance doses of infliximab.38
Despite the positive clinical effects of infliximab, it is associated with adverse effects (Table 3) that may lead to serious and possibly deadly complications.39 Individuals with moderate to severe heart failure, New York Heart Association Functional Class III/IV, should not receive infliximab doses greater than 5 mg/kg, as higher doses are associated with worsening heart failure, increased cardiovascular events, and mortality. Since its introduction in 1998, infliximab has been associated with an increased risk of reactivation of tuberculosis (TB). 40 All patients should be questioned and screened for TB prior to initiating treatment. Serious infections, including the development of sepsis and reactivation of hepatitis B, have been reported. Patients with an active infection should not be started on infliximab until the infection resolves, as some cases of infection resulted in death. Hematological events such as leukopenia, neutropenia, thrombocytopenia, and pancytopenia have also been reported. Thus, signs and symptoms for infection, persistent fevers, and blood dyscrasias should be assessed during and after infliximab therapy. Hypersensitivity reactions such as urticaria, hypotension, dyspnea, fever, rash, myalgias, and facial edema have occurred with the infusions and may be treated with antihistamines, corticosteroids, acetaminophen, or epinephrine. Hepatotoxicity, serum sickness like disease, neurological events, and malignancies are rare yet have been associated with infliximab and were fatal in some cases. Clinicians should determine if the benefits outweigh the risks prior to initiating infliximab treatment for Crohn's disease.
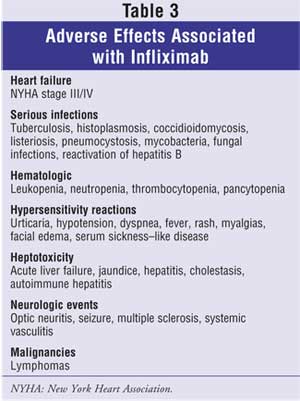
Antibodies known as human antichimeric antibodies (HACA) may develop in patients treated with infliximab. This often leads to delayed hypersensitivity reactions and a loss in therapeutic response. Patients who no longer benefit from infliximab may achieve a response with adalimumab. Adalimumab was first approved for the treatment of rheumatoid arthritis in the U.S. and is now undergoing clinical trials for Crohn's disease. Preliminary data indicate an 80-mg loading dose of adalimumab at baseline and week 2, followed by 40 mg every other week, is well tolerated with a positive clinical response.41,42 Since infliximab is molecularly similar to adalimumab, the adverse effects of this new recombinant human monocolonal immunoglobulin G1 antibody agent may be similar to infliximab, yet this remains to be confirmed. Adalimumab has been associated with an increased risk of reactivation of TB and hepatitis B, thus all patients should be screened prior to initiating treatment. Also, serious infections have been reported; therefore, patients with an active infection should not start treatment until the infection resolves.
Supportive Therapies: Patients with Crohn's disease often have symptoms that may be relieved with adjunctive therapies. Since diarrhea is common in these patients, antidiarrheal agents, including loperamide and diphenoxylate-atropine, may be recommended if there is no risk of intestinal obstruction. Also, pain is a frequent complaint that may require treatment with narcotics. As mentioned earlier, nutritional deficiencies may occur and require vitamin supplementation. The use of probiotics (nonpathogenic microorganisms) and their role in the management of Crohn's disease is inconclusive.43
Surgery: Approximately 75% of Crohn's disease patients will require surgical intervention to alleviate GI symptoms during their lifetime. Unfortunately, surgery is not a cure, as more than 50% of patients relapse by postoperation year 5, and the rate of relapse may be as high as 75% at postoperation year 10. 44 Therefore, surgery should be considered only if the patient has failed all other forms of medical management.
The Role of the Pharmacist
Pharmacists can play an integral role in the medical care of patients with Crohn's disease. They can recommend appropriate over-the-counter adjunctive therapies, such as antidiarrheal agents or nutritional supplements. The pharmacist can counsel patients and educate nurses on how to administer the medications used to treat Crohn's disease, especially as they may come in many different formulations, including tablets, capsules, suppositories, enema, infusions, and/or injections. Additional counseling may be completed regarding the adverse effects associated with the prescribed medications. Recommendations may be made for appropriate regimens to prevent osteopenia and osteoporosis for patients on corticosteroids. Monitoring of immunosuppressant drug levels (e.g., cyclosporine, tacrolimus, TPMT) and evaluating for abnormal laboratory results may benefit the patient's treatment response. Pharmacists can screen and identify significant medical conditions (e.g., TB, congestive heart failure, infections) prior to initiating biological agents such as infliximab or adalimumab. Ultimately, the quality of life of individuals affected with Crohn's disease will improve as pharmacists combine their pharmacotherapeutic and disease state knowledge with a compassionate understanding of patients' needs.
References
1. Kornbluth A, Sachar DB; Practice Parameters Committee of the American College of Gastroenterology. Ulcerative colitis practice guidelines in adults (update): American College of Gastroenterology, Practice Parameters Committee. Am J Gastroenterol. 2004;99:1371-1385.
2. Hanauer SB, Sandborn W; Practice Parameters Committee of the American College of Gastroenterology. Management of Crohn's disease in adults. Am J Gastroenterol. 2001;96:635-643.
3. Loftus EV Jr. Clinical epidemiology of inflammatory bowel disease: Incidence, prevalence, and environmental influences. Gastroenterology. 2004;126:1504-1517.
4. Ekbom A. The epidemiology of IBD: a lot of data but little knowledge. How shall we proceed? Inflamm Bowel Dis. 2004;10 [Suppl 1]:S32-S34.
5. Heresbach D, Alexandre JL, Bretagne JF, et al. Crohn's disease in the over-60 age group: a population based study. Eur J Gastroenterol Hepatol . 2004;16:657-664.
6. Basu D, Lopez I, Kulkarni A, Sellin JH. Impact of race and ethnicity on inflammatory bowel disease. Am J Gastroenterol. 2005;100:2254-2261.
7. Calkins BM. A meta-analysis of the role of smoking in inflammatory bowel disease. Dig Dis Sci. 1989;34:1841-1854.
8. Gibson PR, Shepherd SJ. Personal view: food for thought--western lifestyle and susceptibility to Crohn's disease. The FODMAP hypothesis. Aliment Pharmacol Ther. 2005;21:1399-1409.
9. Fiocchi C. Inflammatory bowel disease: etiology and pathogenesis. Gastroenterology. 1998;115:182-205.
10. Hanauer SB, Sandborn W. Management of Crohn's disease in adults. Am J Gastroenterol.2001;96:635-643.
11. Summers RW, Switz DM, Sessions JT Jr, et al. National Cooperative Crohn's Disease Study: results of drug treatment. Gastroenterology. 1979;77:847-869.
12. Landi B, Anh TN, Cortot A, et al. Endoscopic monitoring of Crohn's disease treatment: a prospective, randomized clinical trial. The Groupe d'Etudes Therapeutiques des Affections Inflammatoires Digestives. Gastroenterology . 1992;102:1647-1653.
13. Steinhart AH, Ewe K, Griffiths AM, et al. Corticosteroids for maintenance of remission in Crohn's disease. Cochrane Database Syst Rev. 2003:CD000301.
14. Bernstein CN, Leslie WD, Leboff MS. AGA technical review on osteoporosis in gastrointestinal diseases. Gastroenterology. 2003;124:795-841.
15. Otley A, Steinhart AH. Budesonide for induction of remission in Crohn's disease. Cochrane Database Syst Rev. 2005 Oct 19:CD000296.
16. Chopra A, Pardi DS, Loftus EV Jr, et al. Budesonide in the treatment of inflammatory bowel disease: the first year of experience in clinical practice. Inflamm Bowel Dis. 2006;12:29-32.
17. Akobeng AK, Gardener E. Oral 5-aminosalicylic acid for maintenance of medically-induced remission in Crohn's Disease. Cochrane Database Syst Rev. 2005:CD003715.
18. Singleton JW, Hanauer SB, Gitnick GL, et al. Mesalamine capsules for the treatment of active Crohn's disease: results of a 16-week trial. Pentasa Crohn's Disease Study Group. Gastroenterology. 1993;104:1293-1301.
19. Messori A, Brignola C, Trallori G, et al. Effectiveness of 5-aminosalicylic acid for maintaining remission in patients with Crohn's disease: a meta-analysis. Am J Gastroenterol. 1994;89(5):692-698.
20. Steinhart AH, Hemphill D, Greenberg GR. Sulfasalazine and mesalazine for the maintenance therapy of Crohn's disease: a meta-analysis. Am J Gastroenterol. 1994;89:2116-2124.
21. Kamm MA. Review article: chronic active disease and maintaining remission in Crohn's disease. Aliment Pharmacol Ther. 2004;20 [Suppl 4]:102-105.
22. Pearson DC, May GR, Fick GH, Sutherland LR. Azathioprine and 6-mercaptopurine in Crohn's disease. A meta-analysis. Ann Intern Med . 1995;123:132142.
23. Pearson DC, May GR, Fick G, Sutherland LR. Azathioprine for maintaining remission of Crohn's disease. Cochrane Database Syst Rev. 2000:CD000067.
24. Aberra FN, Lichtenstein GR. Review article: monitoring of immunomodulators in inflammatory bowel disease. Aliment Pharmacol Ther. 2005;21:307-319.
25. Siegel CA, Sands BE. Review article: practical management of inflammatory bowel disease patients taking immunomodulators. Aliment Pharmacol Ther. 2005;22:1-16.
26. Sun JH, Das KM. Low-dose oral methotrexate for maintaining Crohn's disease remission: where we stand. J Clin Gastroenterol. 2005;39:751-756.
27. Vandeputte L, D'Haens G, Baert F, et al. Methotrexate in refractory Crohn's disease. Inflamm Bowel Dis. 1999;5:11-15.
28. Brynskov J, Freund L, Rasmussen SN, et al. A placebo-controlled, double-blind, randomized trial of cyclosporine therapy in active chronic Crohn's disease. N Engl J Med. 1989;321:845-850.
29. Brynskov J, Freund L, Norby Rasmussen S, et al. Final report on a placebo-controlled, double-blind, randomized, multicentre trial of cyclosporin treatment in active chronic Crohn's disease. Scand J Gastroenterol . 1991;26:689-695.
30. Loftus CG, Egan LJ, Sandborn WJ. Cyclosporine, tacrolimus, and mycophenolate mofetil in the treatment of inflammatory bowel disease. Gastroenterol Clin North Am. 2004;33:141-169.
31. Wild GE. The role of antibiotics in the management of Crohn's disease. Inflamm Bowel Dis. 2004;10:321-323.
32. Sutherland L, Singleton J, Sessions J, et al. Double blind, placebo controlled trial of metronidazole in Crohn's disease. Gut. 1991;32:1071-1075.
33. Prantera C, Berto E, Scribano ML, Falasco G. Use of antibiotics in the treatment of active Crohn's disease: experience with metronidazole and ciprofloxacin. Ital J Gastroenterol Hepatol. 1998;30:602-606.
34. Gionchetti P, Rizzello F, Morselli C, et al. Management of inflammatory bowel disease: does rifaximin offer any promise? Chemotherapy. 2005;51[Suppl]1:96-102.
35. Shafran I, Johnson LK. An open-label evaluation of rifaximin in the treatment of active Crohn's disease. Curr Med Res Opin. 2005;21:1165-1169.
36. Targan SR, Hanauer SB, van Deventer SJ, et al. A short-term study of chimeric monoclonal antibody cA2 to tumor necrosis factor alpha for Crohn's disease. Crohn's Disease cA2 Study Group. N Engl J Med. 1997;337:1029-1035.
37. Nikolaus S, Raedler A, Kuhbacker T, et al. Mechanisms in failure of infliximab for Crohn's disease. Lancet. 2000;356:1475-1479.
38. Hanauer SB, Feagan BG, Lichtenstein GR, et al. Maintenance infliximab for Crohn's disease: the ACCENT I randomised trial. Lancet. 2002;359:1541-1549.
39. Colombel JF, Loftus EV Jr, Tremaine WJ, et al. The safety profile of infliximab in patients with Crohn's disease: the Mayo clinic experience in 500 patients. Gastroenterology. 2004;126:19-31.
40. Keane J, Gershon S, Wise RP, et al. Tuberculosis associated with infliximab, a tumor necrosis factor alpha-neutralizing agent. N Engl J Med . 2001;345:1098-1104.
41. Sandborn WJ, Hanauer S, Loftus EV Jr, et al. An open-label study of the human anti-TNF monoclonal antibody adalimumab in subjects with prior loss of response or intolerance to infliximab for Crohn's disease. Am J Gastroenterol. 2004;99:1984-1989.
42. Papadakis KA, Shaye OA, Vasiliauskas EA, et al. Safety and efficacy of adalimumab (D2E7) in Crohn's disease patients with an attenuated response to infliximab. Am J Gastroenterol. 2005;100:75-79.
43. Meier R, Steuerwald M. Place of probiotics. Curr Opin Crit Care. 2005;11:318-325.
44. Yamamoto, T. Factors affecting recurrence after surgery for Crohn's disease. World J Gastroenterol. 2005;11:3971-3979.
To comment on this article, contact editor@uspharmacist.com.





