US Pharm. 2016;41(12):HS16-HS19.
ABSTRACT: Marijuana, or cannabis, is commonly thought to be a benign substance without adverse effects; however, cannabinoid hyperemesis syndrome (CHS) is a sequela of chronic cannabis use. Clinicians should strongly suspect CHS in long-term users of cannabis who present with intractable nausea and vomiting and whose symptoms are relieved by bathing in hot water. A lack of response to antiemetics should assist in the diagnosis of CHS. The only treatment to successfully resolve CHS is abstinence from cannabis, although haloperidol is being studied as a potential treatment option. A patient experiencing CHS should be counseled to avoid cannabis because continued use will trigger further symptoms.
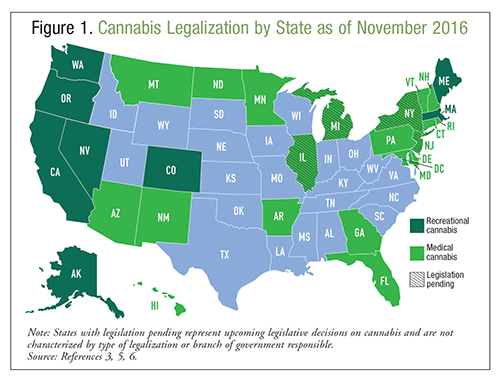
Marijuana, or cannabis, is a natural product derived from a hemp plant known as Cannabis sativa; it is commonly thought of as a benign substance with no adverse effects.1 Cannabis may be smoked, vaporized, or ingested for intoxication or therapeutic benefit, and it has been used for centuries for these purposes. According to the 2014 National Survey on Drug Use and Health, >22 million Americans used cannabis in the preceding month, making this substance the most commonly used illicit drug.2 It is expected that cannabis use in the United States will continue to grow because of increasing legalization rates.3 According to the National Epidemiologic Survey on Alcohol and Related Conditions, residents of states that have legalized medical cannabis are twice as likely to endorse cannabis use as residents of states without legalization.4 Currently, cannabis may be used for medicinal purposes in 28 states, and it also may be used recreationally in eight of them. Refer to FIGURE 1 for information on the legality of cannabis throughout the U.S. at this writing.3,5,6 In addition to legalization, 21 states and the District of Columbia have decriminalized possession of small amounts of cannabis. Medical emergencies related to cannabis use increased by 29% from 2009 to 2011; it is unknown whether this increase was due to the potency of the cannabis, increased legalization, or other factors.1
The human body has an intrinsic cannabinoid system, known as the endocannabinoid system, that contains neurotransmitters (i.e., anandamide) that regulate appetite, sleep, pain, emotion, and movement.7,8 Cannabinoid refers to any compound that affects cannabinoid receptors in the body. Receptors that serve therapeutic purposes include cannabinoid types 1 and 2 (CB1, CB2). CB1 is found in large quantities in the brain and in lesser amounts in spinal and peripheral nervous tissues, whereas CB2 is found in large quantities in peripheral tissues and the immune system.8
Cannabis contains >100 chemicals, of which delta-9-tetrahydrocannabinol (THC) is the most active compound. Because it binds to CB1, THC is responsible for the intoxicating effect of cannabis.1 Other cannabinoids present in cannabis include cannabidiol and cannabigerol, which are not psychoactive. Because of the wide range of activities that the intrinsic system affects, many people have used cannabinoids for intoxication and for therapeutic purposes.
Cannabinoid Hyperemesis Syndrome
Cannabis has been used to promote appetite in the treatment of HIV/AIDS-associated anorexia and wasting and to relieve chemotherapy-induced nausea and vomiting. Although cannabis may be used to treat nausea and vomiting, it may also cause a paradoxical syndrome of nausea and vomiting known as cannabinoid hyperemesis syndrome (CHS). CHS is a clinical diagnosis characterized by chronic cannabis use.9 A unique feature of CHS is cyclic nausea and vomiting that may be unresponsive to standard antiemetic therapy but responsive to compulsive hot bathing.3,9 The first case report documenting CHS (2004) described nine patients in Australia. Chronic cannabis use occurred prior to presentation, and symptoms included nausea and vomiting. Most patients reported that they bathed in hot water to alleviate symptoms. Cessation of cannabis use led to resolution of illness, whereas patients who continued to use cannabis remained ill.10 Since this time, additional case reports and series have been published that further describe CHS; however, no randomized, controlled studies have evaluated or defined it.
In Colorado, rates of CHS have doubled since 2009, when cannabis was legalized there.3 However, it is important to note that CHS often goes unrecognized by clinicians, which may lead to an extensive and costly patient workup. Diagnosis can be challenging, and CHS may be misdiagnosed as many other disorders, including cyclic vomiting syndrome (CVS). Although some patients who use marijuana present with CVS, there are several differentiating factors between CVS and CHS. CVS usually involves a personal or family history of migraines, a history of psychiatric disorders, and a lack of compulsive hot bathing.11 Patients with CHS often present to the emergency department (ED) because of symptoms. A case report by Soriano-Co and colleagues demonstrated that patients diagnosed with CHS were more likely to have frequent clinician visits, ED visits, and hospitalizations.12 On average, a patient had five visits to a clinician, seven visits to the ED, and three hospitalizations before diagnosis occurred, indicating that patients with CHS could be seen in any care setting.12 Further complicating diagnosis is the use of synthetic cannabinoids, such as K2, aka Spice, which cannot be detected in urine drug screens.13 CHS must be differentiated from other disorders that cause nausea and vomiting, and the diagnosis is generally one of exclusion.
Pathophysiology
There are multiple hypotheses regarding the pathophysiology of CHS; however, because of the multitude of active chemicals in cannabis and the existence of various sites of action, the definitive pathophysiological process is unknown. Additionally, not all cannabis users develop CHS, causing further difficulty in describing the syndrome.
At low doses, THC is known to exert an antiemetic effect, but with heavy and chronic cannabis use, the opposite is seen. It has been suggested that THC accumulates over time because of its large volume of distribution. THC may directly activate CB1 receptors in the enteric nervous system and reduce gastric motility, increasing the risk of nausea and vomiting with excessive activation.9 Synthetic cannabinoids, which mimic both the effect and the structure of THC, are hypothesized to cause overstimulation of the CB1 receptor because of their potent agonist effects.14 These CB1 receptors are also found in the hypothalamus and may induce emesis by impairing thermoregulation, leading to the potential relief of symptoms through hot bathing.9,11
Risk Factors, Clinical Course, and Diagnosis
Chronic cannabis use is the main risk factor for the development of CHS. A patient’s risk of CHS increases with prolonged use of cannabis.9 Daily cannabis use appears to be more likely to cause CHS than less frequent use; however, any amount of cannabis can lead to CHS.15
CHS has several phases, which have been classified as preemetic, hyperemetic, and recovery.9,16 The preemetic phase, which can last for months to years, is characterized by nausea, fear of vomiting, and abdominal discomfort. Commonly, cannabis users increase their cannabis intake during the preemetic phase in an effort to alleviate nausea.11 During the hyperemetic phase, which typically lasts 24 to 48 hours, the patient experiences frequent nausea and vomiting that is severe in nature. This may lead to weight loss and acute dehydration, potentially resulting in prerenal failure.17,18 The persistent nausea and vomiting can cause electrolyte abnormalities to develop.
To alleviate symptoms, patients may start hot bathing—i.e., remaining in a hot bath or shower for hours.11 This is a self-learned behavior that becomes compulsive once the patient realizes the benefit. Hot bathing may help with thermoregulation and reduction of blood flow to the stomach by affecting peripheral vasodilation and redistribution from splanchnic circulation, resulting in decreased vomiting.11 Unfortunately, hot bathing may increase the risk of dehydration and acute renal failure.17 Hot bathing should be considered a pathognomonic feature, as it is not seen with other conditions that may have an otherwise similar presentation. Patients typically discontinue hot bathing when symptoms are not present and begin it again once nausea and vomiting recur after future cannabis use.
Many patients seek medical care for their symptoms during the hyperemetic phase. As a result, patients may undergo extensive negative workups for various other conditions, such as CVS, pancreatitis, and gastroparesis, which may mimic some symptoms of CHS. Once the nausea and vomiting subside, usually within 48 hours, the patient enters the recovery phase. Recovery may range from days to months and is associated with cessation of cannabis use. If the patient reinitiates cannabis, symptoms usually return.11
It is important to consider the impact of CHS on a patient’s other disease states. There are case reports of patients with CHS who developed CHS-related medical problems other than renal failure. Gregoire and colleagues described a patient with a history of bipolar mania and cannabis use who developed CHS.19 The patient attributed the intractable vomiting to lithium and stopped taking the lithium, and a manic episode occurred. Reinitiation of lithium in the hospital triggered no additional vomiting, indicating that cannabis was the likely cause.19
Diagnostic criteria for CHS have been proposed. Long-term cannabis use is essential for diagnosis. The length of time it takes to develop CHS varies; however, most patients present 1 to 5 years after chronic cannabis use. Presentation with symptoms similar to those of CHS prior to 1 year of chronic cannabis use should not preclude diagnosis. Major features of CHS include severe cyclic nausea and vomiting, resolution of symptoms upon cannabis cessation, relief achieved by bathing in hot water, epigastric or periumbilical abdominal pain, and weekly cannabis use. Supportive features leading to a diagnosis of CHS include age <50 years, weight loss >5 kg, morning symptom occurrence, normal bowel habits, and normal findings on other workups.15 TABLE 1 summarizes common findings supporting a diagnosis of CHS.14

Potential Treatments
Supportive care is indicated for all patients who present in the hyperemetic phase of CHS. If the patient can tolerate it, oral intake of fluids is recommended for hydration. Some patients cannot tolerate oral hydration owing to excessive vomiting, so IV fluids may be indicated for volume depletion associated with dehydration.11 Because electrolyte disturbances may occur with vomiting, monitoring and replacement of electrolytes is indicated.
Compulsive hot bathing is a hallmark learned behavior that patients may use to minimize symptoms associated with CHS. As was discussed previously, hot bathing may help with thermoregulation. Another theory is that peripheral vasodilation and redistribution from splanchnic circulation may reduce blood flow to the stomach, resulting in decreased vomiting.11 A single case report has indicated that the alleviation of symptoms by hot bathing may wane over time; however, this phenomenon appears to be uncommon.20
Antiemetics have been used without success to relieve symptoms of CHS. Lack of response to antiemetics should cause the clinician to suspect CHS. In case reports, patients have had no therapeutic relief from nausea and vomiting with ondansetron, promethazine, chlorpromazine, or metoclopramide.21 In a small case series, 87.5% of patients did not respond to standard antiemetic therapy.12
Haloperidol exerts antipsychotic effects by antagonizing dopamine D2 receptors in the mesolimbic and mesocortical pathways. Haloperidol is traditionally used to treat agitation; however, it has been used successfully as an antiemetic in general surgery and oncology. D2 receptors are also present in the chemoreceptor trigger zone, which may account for these antiemetic properties. Animal studies suggest that CB1 may be affected by haloperidol, rendering this agent a potential treatment option.22,23 Witsil and Mycyk evaluated this hypothesis retrospectively in four patients with nausea and vomiting refractory to standard antiemetic therapy.21 All patients found relief within 1 to 2 hours of receiving IV haloperidol 5 mg.21,24 This small case study demonstrates that haloperidol may relieve CHS-related nausea and vomiting sufficiently to prevent hospital admission, which may lead to cost savings for the healthcare system.19 The risk of QTc prolongation with haloperidol must be considered, especially in patients with electrolyte disturbances as a result of excessive vomiting. Additional studies are warranted to determine haloperidol’s place in therapy and its optimal dosing.
Cessation of cannabis is the only treatment that relieves and prevents symptoms associated with CHS. In the study that first described CHS, abstinence from cannabis resulted in cessation of nausea and vomiting in seven of 10 patients, whereas the others continued to suffer ongoing illness. Three of the seven patients later rechallenged themselves with cannabis, and symptoms returned within a few months.10 These findings have been replicated in recent case reports.12,25 Patients should be educated about cannabis cessation as a symptom reliever, and resources to assist with cessation should be provided.
Conclusion
As cannabis is legalized in more states, it is likely that its use will increase and more cases of CHS will be reported. Clinicians should suspect CHS in patients who present with a history of cannabis or synthetic cannabinoid use and symptoms including intractable nausea and vomiting, along with reports of the use of hot bathing for symptom control. This conjecture could prevent extensive and costly workups for other conditions. Pharmacists have a role in counseling patients about the risks associated with cannabis use and referring patients with CHS symptoms for appropriate follow-up care. Case reports in the literature have identified risk factors for CHS. Unfortunately, CHS is relatively underreported, and its exact mechanism is unknown. Diagnosis and treatment of CHS is currently based on small case reports. Information should continue to be evaluated as it becomes available in order to ensure appropriate treatment.
REFERENCES
1. National Institute on Drug Abuse (NIDA). What is marijuana? www.drugabuse.gov/publications/research-reports/marijuana/what-marijuana. Accessed August 25, 2016.
2. NIDA. What is the scope of marijuana use in the United States? www.drugabuse.gov/publications/research-reports/marijuana/what-scope-marijuana-use-in-united-states. Accessed August 25, 2016.
3. Kim HS, Monte AA. Colorado cannabis legalization and its effect on emergency care. Ann Emerg Med. 2016;68:71-75.
4. Cerdá M, Wall M, Keyes KM, et al. Medical marijuana laws in 50 states: investigating the relationship between state legalization of medical marijuana and marijuana use, abuse and dependence. Drug Alcohol Depend. 2012;120:22-27.
5. CNN Money. Where pot is legal. http://money.cnn.com/interactive/news/economy/marijuana-legalization-map/index.html. Accessed August 25, 2016.
6. Marijuana Policy Project. 2016 marijuana policy reform legislation. www.mpp.org/states/key-marijuana-policy-reform. Accessed November 13, 2016.
7. NIDA. How does marijuana produce its effects? www.drugabuse.gov/publications/research-reports/marijuana/how-does-marijuana-produce-its-effects. Accessed August 2016.
8. Manzanares J, Julian MD, Carrascosa A. Role of the cannabinoid system in pain control and therapeutic implications for the management of acute and chronic pain episodes. Curr Neuropharmacol. 2006;4:239-257.
9. Galli JA, Sawaya RA, Friedenberg FK. Cannabinoid hyperemesis syndrome. Curr Drug Abuse Rev. 2011;4:
241-249.
10. Allen JH, de Moore GM, Heddle R, Twartz JC. Cannabinoid hyperemesis: cyclical hyperemesis in association with chronic cannabis abuse. Gut. 2004;53:1566-1570.
11. Ruffle JK, Bajgoric S, Samra K, et al. Cannabinoid hyperemesis syndrome: an important differential diagnosis of persistent unexplained vomiting. Eur J Gastroenterol Hepatol. 2015;27:1403-1408.
12. Soriano-Co M, Batke M, Cappell MS. The cannabis hyperemesis syndrome characterized by persistent nausea and vomiting, abdominal pain, and compulsive bathing associated with chronic marijuana use: a report of eight cases in the United States. Dig Dis Sci. 2010;55:3113-3119.
13. Ukaigwe A, Karmacharya P, Donato A. A gut gone to pot: a case of cannabinoid hyperemesis syndrome due to K2, a synthetic cannabinoid. Case Rep Emerg Med. 2014;2014:167098.
14. Hopkins CY, Gilchrist BL. A case of cannabinoid hyperemesis syndrome caused by synthetic cannabinoids. J Emerg Med. 2013;45:544-546.
15. Simonetto DA, Oxentenko AS, Herman ML, Szostek JH. Cannabinoid hyperemesis: a case series of 98 patients. Mayo Clin Proc. 2012;87:114-119.
16. Lu ML, Agito MD. Cannabinoid hyperemesis syndrome: marijuana is both antiemetic and proemetic. Cleve Clin J Med. 2015;82:429-434.
17. Habboushe J, Sedor J. Cannabinoid hyperemesis acute renal failure: a common sequela of cannabinoid hyperemesis syndrome. Am J Emerg Med. 2014;32:690.e1-e2.
18. Srihari P, Liu M, Punzell S, et al. Cannabinoid hyperemesis syndrome associated with compulsive showering and acute kidney injury. Prim Care Companion CNS Disord. 2016;18(1).
19. Gregoire P, Tau M, Robertson D. Cannabinoid hyperemesis syndrome and the onset of a manic episode. BMJ Case Rep. 2016;2016.
20. Bagdure S, Smalligan RD, Sharifi H, Khandheria B. Waning effect of compulsive bathing in cannabinoid hyperemesis. Am J Addict. 2012;21:184-185.
21. Witsil JC, Mycyk MB. Haloperidol, a novel treatment for cannabinoid hyperemesis syndrome. Am J Ther. 2014 Nov 12 [Epub ahead of print].
22. Schulze DR, Carroll FI, McMahon LR. Interactions between dopamine transporter and cannabinoid receptor ligands in rhesus monkeys. Psychopharmacology (Berl). 2012;222:425-438.
23. Desai RI, Thakur GA, Vemuri VK, et al. Analysis of tolerance and behavioral/physical dependence during chronic CB1 agonist treatment: effects of CB1 agonists, antagonists, and noncannabinoid drugs. J Pharmacol Exp Ther. 2013;344:319-328.
24. Hickey JL, Witsil JC, Mycyk MB. Haloperidol for treatment of cannabinoid hyperemesis syndrome. Am J Emerg Med. 2013;31:1003.e5-e6.
25. Pélissier F, Claudet I, Gandia-Mailly P, et al. Cannabis hyperemesis syndrome in the emergency department: how can a specialized addiction team be useful? A pilot study. J Emerg Med. 2016;51:544-551.
To comment on this article, contact rdavidson@uspharmacist.com.





