ABSTRACT: Opioid analgesics are the mainstay treatment of moderate-to-severe pain. However, opioids carry a risk of misuse, abuse, and death, and recently have become a public health challenge. In response to the steady increase in opioid abuse, pressure has been growing on pharmaceutical companies to develop prescription opioid formulations that help deter abuse while remaining readily accessible for pain management. This article discusses some of the novel abuse-deterrent formulation methods, including aversion, physical barrier, and agonist-antagonist. It also addresses some of the legislative initiatives and the role of pharmacists in providing education to health care professionals and patients as well as increasing their awareness of prescription opioid misuse and abuse.
Pain is a significant public health problem in the United States. It is estimated that over 100 million people in the U.S. live with chronic pain, a prevalence that is greater than heart disease, diabetes, and cancer combined.1 The consequences of unrelieved pain are associated with reduced quality of life and are estimated to cost society over $635 billion annually.1 Pain is recognized as a disorder, and is associated with many psychosocial conditions, including depression, decreased appetite, and insomnia.2
Prevalence of Opioid Abuse
Prescription opioid analgesics are the mainstay of pharmacologic management of pain. They are administered through various routes and are available in many dosage forms, some of which are novel. Over the last few years, there has been a debate regarding the long-term use of opioids in the treatment of chronic nonmalignant pain. This discussion stems from ongoing concerns about adverse effects, lack of long-term data, and the potential for opioid misuse and abuse.
In the past two decades, the use of opioid therapy for the treatment of pain has dramatically increased. Simultaneously, opioid prescription abuse and overdose has markedly increased.3,4 From 1997 to 2007, the milligram-per-person use of prescription opioids in the U.S. increased from 74 to 369 mg, an increase of 402%.3 In addition, in 2000, retail pharmacies dispensed 174 million prescriptions for opioids; by 2009, 257 million prescriptions were dispensed, an increase of 48%.5 National surveys show that opioid misuse has increased dramatically over the past decade and that opioid medications have surpassed cocaine and heroin as the leading drugs of abuse.6,7
Risk Evaluation and Mitigation Strategy
Due to the emerging issues of opioid misuse and abuse, the FDA issued a new Risk Evaluation and Mitigation Strategy (REMS) for extended-release (ER) and long-acting (LA) opioids in July 2012.8 The REMS program stems from the national prescription drug abuse plan that was announced by the Obama administration in 2011.8 REMS is a risk management plan that exceeds standard drug prescribing information; the FDA selected the ER and LA formulations of opioids because of the inherent risks of using these drugs. These formulations contain greater amounts of drug compared to the short-acting formulations, thus making those medications more dangerous in situations of abuse and misuse. The program focuses on educating providers and patients on the safe use of ER and LA opioids while ensuring that patients who require treatment with opioids have access to them. Manufacturers are responsible for creating educational programs and materials for all Drug Enforcement Administration (DEA)–registered prescribers.
Prescribers’ education is an essential element of the REMS program; it encompasses knowledge about the drug, patient assessment, initiation, modification or discontinuation of therapy, management and monitoring of patients, and providing counseling. Furthermore, prescribers are encouraged to use assessment tools to stratify risk of addiction in patients receiving opioid prescription drugs.9 Pharmacists will distribute an updated Medication Guide with ER/LA opioid prescriptions instructing patients to consult their health care providers prior to modifying doses, describing signs of overdose, and giving emergency contact information and recommendations for safe storage to prevent accidental ingestion. The FDA hopes to train 25% of the 320,000 prescribers in the United States within 1 year, 50% after 2 years, and 60% within 4 years. Follow-up surveys will be conducted to assess prescriber understanding and to determine if the program is negatively affecting patient access.7,9
Most recently, in an effort to further safety precautions with the use of ER/LA opioid analgesics, the FDA has imposed safety labeling changes and postmarket study requirements. The labeling changes will include an updated indication emphasizing the use of ER/LA opioids in patients with pain severe enough to require daily, around-the-clock, long-term opioid treatment and for which alternative treatment is inadequate. In addition, the FDA is requiring a new boxed warning on ER/LA opioids analgesics to caution that chronic maternal use of these products during pregnancy can result in neonatal opioid withdrawal syndrome (NOWS).10
Prescription Drug Monitoring Programs
Prescription Drug Monitoring Programs (PDMPs or PMPs) are statewide electronic databases that collect prescribing and dispensing data on controlled substances dispensed in the state. These programs aim to reduce prescription drug abuse and diversion. The benefits of PDMPs are multifactorial in that they help to provide patient care, act as a warning system, and monitor prescribing to prevent diversion. Licensing boards can utilize this system to track prescribing habits and deter diversion. Each state has its own rules and regulations that govern PDMPs.11,12
Abuse-Deterrent Formulations (ADFs)
The pharmaceutical industry recognizes the growing problem that accompanies the increased use of opioids and has set out to create unique compounds that prevent abuse and diversion. This step has spurred the development of abuse-deterrent and abuse-resistant formulations of opioids.13 Current innovations focus on the development of drug delivery systems that are resistant to abuse and diversion. Pharmaceutical developers are experimenting with adding active ingredients or excipients, making the active ingredient(s) unavailable for unintended routes of administration, and preventing the modification of the drug for a faster onset of action. These new special formulations have the potential to reduce the public health burden of prescription opioid abuse. Several different types of ADFs have emerged, including aversion, physical barrier to tampering, and agonist-antagonist combination (TABLE 1).
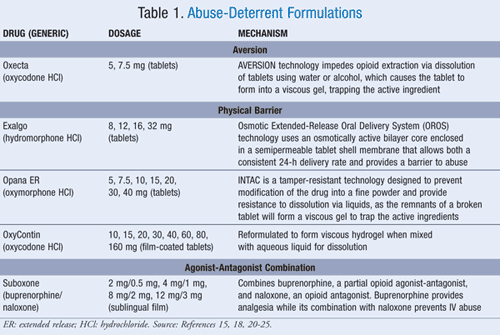
Aversion: One approach to deter abuse potential is the addition of substances with opioid products that produce some type of unpleasant effect in patients who ingest tampered products. Acura Pharmaceuticals has been developing abuse-deterrent products for nearly 10 years using the patented abuse-deterrent AVERSION technology.14 This technology impedes opioid extraction via dissolution of tablets using water or alcohol, which causes the tablet to form into a viscous gel. The active ingredient is then trapped in the gel and becomes difficult to draw up into a syringe for injection. Furthermore, the delivery system also discourages nasal snorting of crushed tablets by including excipients that can cause nasal irritation.14
Oxecta (oxycodone hydrochloride [HCl]), a new formulation of oxycodone that implements AVERSION technology, was approved in 2011.15 Available as 5- and 7.5-mg tablets, Oxecta is indicated for the management of moderate-to-severe pain. When crushed, the tablet breaks down into chunks instead of powder. Furthermore, it is not viable as an injection because when mixed with liquids, it becomes “sudsy.”16 Oxecta contains sodium lauryl sulfate, which will cause discomfort to the nasal passage if snorted.15 The manufacturer has recommended that Oxecta not be given via nasogastric, gastric, or other feeding tubes as it may cause obstruction. Thus, Oxecta may only be appropriate in patients who can tolerate oral therapy.
Physical Barrier: Other common methods of tampering with prescription opioids include physical manipulations (e.g., crushing) and chemical manipulation (e.g., dissolving in alcohol). In 2008, the U.S. Substance Abuse and Mental Health Services Administration’s National Survey on Drug Use and Health reported the number of nonmedical users of OxyContin (controlled-release oxycodone) aged ≥12 years was approximately half a million.17
In response to the increased abuse of OxyContin, the FDA approved a new controlled-release oxycodone formulation in spring 2010.18 The new version was reformulated with physical and chemical properties that make abuse via the intranasal route and injection difficult. The reformu-lated tablets contain polyethylene oxide (PEO). PEO serves as an abuse deterrent because it is a nonionic hydrophilic polymer that is insoluble in alcohol. Upon contact with water, it forms a viscous gel. Attempts to dissolve the tablets in liquid result in a gummy substance that cannot be injected or snorted. The new formulation is also harder to break, cut, chew, or crush. The tablets either do not break or break into pieces that retain some controlled-release functionality. According to the new package insert labeling approved by the FDA in April 2013, in vitro physical and chemical tablet manipulation studies of the new formulation show that it resists crushing, breaking, and dissolution using various tools and solvents.18
Another example of physical barrier is Opana ER (oxymorphone HCl), which is indicated for the relief of moderate-to-severe pain in patients requiring around-the-clock opioid treatment for an extended period of time.19 It is available in 5-, 7.5-, 10-, 15-, 20-, 30-, and 40-mg tablets. This formulation uses INTAC technology created by Grüenthal. INTAC is a tamper-resistant technology designed to prevent modification of the drug into a fine powder. The technology for the ER formulation incorporates PEO, which has a high molecular weight, and uses a “hot-melt extrusion process.” This process combines heat and pressure so that tablets have mechanical strength that resists crushing.20 Furthermore, if the delivery system is compromised, INTAC also provides resistance to dissolution via liquids, as the remnants of a broken tablet will form a viscous gel to trap the active ingredients.21
Exalgo (hydromorphone HCl) is indicated as once-daily administration for the management of moderate-to-severe pain in opioid-tolerant patients. It is available as 8-, 12-, 16-, and 32-mg tablets. Exalgo uses the Osmotic Extended-Release Oral Delivery System (OROS).22 This technology has an osmotically active bilayer core enclosed in a semipermeable tablet shell membrane that allows both a consistent 24-hour delivery rate and provides a barrier to abuse.23
Agonist-Antagonist Combination: Several formulation products containing an opioid agonist in combination with an antagonist are already on the market. Suboxone (buprenorphine HCl/naloxone HCl) is indicated for treatment of opioid dependence and is available as a sublingual film in strengths of 2 mg/0.5 mg, 4 mg/1 mg, 8 mg/2 mg, and 12 mg/3 mg.24 This drug uses a different method to deter abuse. Suboxone combines buprenorphine, a partial opioid agonist-antagonist, and naloxone, an opioid antagonist. Buprenorphine provides analgesia while its combination with naloxone prevents IV abuse. The naloxone component is not sublingually absorbed and has minimal oral absorption; therefore, it does not exert its antagonist effect when taken orally or sublingually. However, if Suboxone is crushed and then injected IV or IM, the naloxone component will be directly introduced to the systemic circulation, where it will antagonize the effects of buprenorphine. This antagonistic action, produced by the naloxone contained in Suboxone, causes opioid-dependent patients to experience withdrawal, thereby deterring abuse.24,25
Pipeline Abuse-Deterrent Formulations
Acura Pharmaceuticals has multiple pipeline products in development with its patented abuse-deterrent AVERSION technology. Acuracet, a combination of oxycodone HCl and acetaminophen, is currently in the formulation and stability stages of development. Vycavert, a combination of hydrocodone bitartrate and acetaminophen, is in the clinical and laboratory testing phases of development. Both are immediate-release opioids with a proposed indication for relief of moderate-to-severe pain, designed to limit abuse via nasal snorting and injection.26,27
Naloxone Access Law
With the steady increase in fatal drug overdoses related to opioid use, many states have amended laws to allow for access to emergency care and treatment of opioid overdose. As an opioid antagonist, naloxone is essential for the reversal of an overdose. Overdoses often occur when a victim is with friends or family; therefore, if these people can act quickly and administer naloxone, they may be able to save a victim’s life. Naloxone access and “Good Samaritan” laws remove the possibility of negative legal action against prescribers who act in good faith to prescribe naloxone to those who may benefit from it. They also encourage bystanders to become Good Samaritans by summoning emergency responders without fear of arrest or unfavorable legal consequences.28
Role of the Pharmacist
Pharmacists are in a unique position to provide education regarding opioid use in both inpatient and outpatient settings. Pharmacists are valuable members of the interdisciplinary team because of their knowledge of opioid medications, their ability to perform a comprehensive review of past and current pharmacologic interventions for pain, and their ability to provide valuable insight regarding the most appropriate opioids for pain management. The implementation of PDMPs and the REMS program opens up the opportunity for pharmacists to be further involved, such as in tracking prescriptions and alerting providers of potential abuse or illegal prescription filling. Pharmacists can educate providers and patients on the use of opioids, help prevent their abuse and overuse, and reduce the potential for addiction and dependence. Pharmacists can also emphasize to patients the techniques for proper opioid use, storage, and disposal. With the newer formulations on the market, pharmacists can educate both health care providers and patients about types of abuse-deterrent formulations and their advantages. Overall, education by pharmacists can help to decrease the potential for opioid abuse.
Conclusion
As the use of opioids reaches an all-time high, concern for their misuse and abuse rises simultaneously. In response to the clinical concerns about drug abuse, many manufacturers are developing opioid products that have abuse-deterrent qualities. The pharmaceutical industry has begun to offer solutions and will likely continue to innovate and design new methods to address this concern. The new technologies described here and future improvements will give health care providers the means to address pain management while mitigating the concern for and rise of opioid abuse and misuse. It is imperative that clinicians understand the characteristics of these agents if they are to improve the care of their patients with pain.
REFERENCES
1. Institute of Medicine. Relieving Pain in America: A Blueprint for Transforming Prevention, Care, Education, and Research.
Washington, DC: National Academies of Science; 2011.
www.iom.edu/Reports/2011/Relieving-Pain-in-America-A-Blueprint-for-transforming-Prevention-Care-Education-Research.aspx.
Accessed June 19, 2013.
2. Argoff CE, Albrecht P, Irving G, Rice F. Multimodal analgesia for chronic pain: rationale and future directions. Pain Med. 2009;10(suppl 2):S53-S66.
3. Manchikanti L, Fellows B, Ailinani H, et al. Therapeutic use, abuse, and nonmedical use of opioids: a ten-year perspective. Pain Physician. 2010;13:401-435.
4. Toblin RL, Paulozzi LJ, Logan JE, et al. Mental illness
and psychotropic drug use among prescription drug overdose deaths: a
medical examiner chart review. J Clin Psychiatry. 2010;71:491-496.
5. Joint meeting of the Anesthetic and Life Support Drugs
Advisory Committee and the Drug Safety and Risk Management Advisory
Committee. Risk Evaluation and Mitigation Strategies (REMS) for
extended-release and long-acting opioid analgesics. July 22-23, 2010.
www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/Drugs/AnestheticAndLifeSupportDrugsAdvisoryCommittee/UCM217510.pdf.
Accessed September 9, 2013.
6. Paulozzi LJ, Budnitz DS, Xi Y. Increasing deaths from opioid analgesics in the United States. Pharmacoepidemiol Drug Saf. 2006;15:618-627.
7. Kuehn BM. Opioid prescriptions soar: increase in legitimate use as well as abuse. JAMA. 2007;297:249-251.
8. Questions and answers: FDA approves a Risk Evaluation
and Mitigation Strategy (REMS) for extended-release and long-acting
opioid analgesics. July 9, 2012.
www.fda.gov/Drugs/DrugSafety/InformationbyDrugClass/ucm309742.htm.
Accessed June 12, 2013.
9. Extended-release (ER) and long-acting (LA) opioid
analgesics Risk Evaluation and Mitigation Strategy (REMS). FDA. April
2013.
www.fda.gov/downloads/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/UCM311290.pdf.
Accessed June 12, 2013.
10. FDA announces safety labeling changes and post market
study requirements for extended-release and long-acting opioid
analgesics. FDA news release. September 10, 2013.
www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm367726.htm.
Accessed September 23, 2013.
11. Questions and answers: state prescription drug
monitoring programs. DEA. October 2011.
www.deadiversion.usdoj.gov/faq/rx_monitor.htm. Accessed June 24, 2013.
12. Prescription drug monitoring programs. Office of
National Drug Control Policy. April 2011.
https://www.ncjrs.gov/pdffiles1/ondcp/pdmp.pdf. Accessed June 24, 2013.
13. Schneider JP, Matthews M, Jamison RN. Abuse-deterrent
and tamper-resistant opioid formulations: what is their role in
addressing prescription opioid abuse? CNS Drugs. 2010;24:805-810.
14. AVERSION technology. Acura Pharmaceuticals, Inc.
http://acurapharm.com/research-development/aversion-technology. Accessed
June 12, 2013.
15. Oxecta (oxycodone HCl) package insert. Bristol, TN: King Pharmaceuticals; June 2011.
16. Fiore K. FDA okays new abuse-resistant opioid. MedPage Today. June 20, 2011. www.medpagetoday.com/ProductAlert/Prescriptions/27157. Accessed June 12, 2013.
17. Substance Abuse and Mental Health Services Administration (SAMHSA). Results From the 2008 National Survey on Drug Use and Health: National Findings. Office of Applied Studies, NSDUH Series H-36, HHS Publication No. SMA 09-4434. Rockville, MD: SAMHSA; 2009.
18. OxyContin (oxycodone HCl) package insert. Stamford, CT: Purdue Pharma LP; June 2013.
19. Opana ER (oxymorphone HCl) package insert. Lincoln, NE: Novartis Consumer Health; July 2012.
20. INTAC. Grüenthal’s TRF technology.
www.grunenthal.com/cms/cda/_common/inc/display_file.jsp?fileID=215700010.
Accessed June 12, 2013.
21. INTAC formulation technology protects intended drug
action. Grüenthal. www.intac.grunenthal.com/grt-intac/212000676.jsp.
Accessed June 12, 2013.
22. Exalgo (oxymorphone HCl) package insert. Hazelwood, MO: Mallinckrodt Brand Pharmaceuticals; November 2010.
23. Sathyan G, Zu E, Thipphawong J, et al. Pharmacokinetic
investigation of dose proportionality with a 24-hour controlled-release
formulation of hydromorphone. BMS Clin Pharmacol. 2007;7:1-8.
24. Suboxone (buprenorphine and naloxone) package insert. Richmond, VA: Reckitt Benckiser Pharmaceuticals; August 2012.
25. Orman JS, Keating GM. Buprenorphine/naloxone: a review of its use in the treatment of opioid dependence. Drugs. 2009;69:577-607.
26. Acuracet Tablets. Acura Pharmaceuticals, Inc. http://acurapharm.com/products/acuracet-tablets/. Accessed June 12, 2013.
27. Vycavert Tablets. Acura Pharmaceuticals, Inc. http://acurapharm.com/products/vycavert-tablets/. Accessed June 12, 2013.
28. Legal interventions to reduce overdose mortality:
naloxone access and overdose good samaritan laws. The Network for Public
Health Law. June 2013.
www.networkforphl.org/_asset/qz5pvn/network-naloxone-10-4.pdf. Accessed
June 12, 2013.
To comment on this article, contact rdavidson@uspharmacist.com.





