US Pharm. 2012;37(1):42-45.
Multiple sclerosis (MS) is an immune-mediated disease of the central nervous system (CNS). It is clinically characterized by recurrent or progressive neurologic dysfunction caused by multiple demyelinated CNS lesions, which occur in the brain, spinal cord, and optic nerve. Symptom onset is from 20 to 50 years of age. The disease can be limited to infrequent episodes of acute neurologic symptoms or may become chronic, relapsing, or progressive and show cumulative neurologic disability over 10 to 20 years.1
In 2008, Alonso and Hernán reviewed the available data on MS incidence rates and geographic location and found that women had the highest overall incidence rate at 3.6 cases as compared to men at 2 cases per 100,000 person-years.2 In the past few decades, the highest prevalence of MS was associated with higher latitudes geographically and with people of northern European descent, especially Scandinavians. This may be changing, possibly due to migration of members of this ethnic group to areas below the 40° latitude.2 In those of northern European ancestry and with a familial link to MS, moving to the southern regions seems to reduce the risk of developing MS.1
Symptoms and Diagnosis
The etiology of MS is unknown; it is often a diagnosis of exclusion based on symptoms, age, and MRI findings. The typical clinical findings include optic neuritis, internuclear ophthalmoplegia, heat sensitivity, and Lhermitte’s sign.1 In 20% of patients with MS, optic neuritis is the initial symptom and up to 50% of patients will eventually develop this symptom. In patients with optic neuritis, vision disturbances include unilateral vision loss and retrobulbar pain with eye movement. Often, the funduscopic eye examination will be normal in early stages of the disease. Diplopia may be caused by internuclear ophthalmoplegia, paralysis of the ocular muscles. The eye does not move correctly to the center when the patient looks left or right (lateral gaze) but moves to the center if the patient looks at a central point (“crossed eyes”). Patients with MS may find that a hot bath, spicy foods, or exercise causes an exacerbation of symptoms. When the body temperature returns to normal, the symptoms resolve. Lhermitte’s sign is when forward head flexion causes sudden electric-like shocks down the spine or arms. This is reproducible and may indicate a cervical lesion. Other less specific symptoms include fatigue, weakness, sensory loss, ataxia, and bowel and bladder incontinence.1
McDonald et al first added MRI criteria to the diagnostic pattern. The criteria specify the number of evident lesions and their location; this was updated in 2005 to demonstrate progression within 30 or more days.3 A revised set of diagnostic criteria is proposed by MAGNIMS (European MS MRI consortium). This group’s findings allow a diagnosis with only one additional lesion in at least two of four characteristic locations on the brain or spinal cord, potentially with only one MRI.4 Nonspecific but associated cerebrospinal fluid (CSF) findings include mildly elevated protein (<100 mg/dL), slight increase in monocytes (<50 cells/µL), and abnormal synthesis of immunoglobulin.1 No pathognomonic biomarkers are currently available for blood or urine.
Types of MS
MS is usually classified into four types that may not be totally distinct. Relapsing remitting MS (RRMS) is the most common, affecting nearly 85% of diagnosed patients.1 The patients in this category have periods of no or lessened symptoms and then will have worsening of symptoms that they have had before or new symptoms over a 48-hour period with no fever. Secondary progressive MS (SPMS) is when patients with RRMS move into a progressive course of their disease with few, if any, periods of lessened symptoms. When patients convert to SPMS, neurologic disability seems to be most devastating. Primary progressive MS (PPMS) is seen in approximately 10% of patients. Patients decline steadily over time without a relapse period. Progressive relapsing MS (PRMS) is the rarest type, affecting 5% of patients. Patients have a steady disease progression with periods of acute exacerbations of symptoms.1
Prognosis of early onset of disability may be predicted early in the course of the disease. Prior to the availability of disease-modifying agents (DMAs), a systemic review of the literature for patients with RRMS revealed that incomplete recovery from the first episode, a short interval between the initial and a subsequent symptomatic episode, or early episodes that involved bowel or bladder sphincter dysfunction, were associated with the highest rate and form of disability. Relapse frequency did not correlate with long term disability prediction.5 In the United Kingdom, Fisniku et al found that lesion volume progression shown on MRI during the early development of MS (0-5 years) is correlated with increased disability after 20 years as determined by the Expanded Disability Status Scale (EDSS) and Multiple Sclerosis Functional Composite (MSFC) score.6 In studied patients who developed SPMS, lesion volume and the rate of lesion growth were much higher than in those who remained as RRMS patients.6
The EDSS is a method of quantifying disability in MS. This method quantifies disability in eight Functional Systems (FS) and allows neurologists to assign a Functional System Score (FSS) in each of these. The FS are pyramidal, cerebellar, brainstem, sensory, bowel and bladder, visual, cerebral, and other. Patients with MS who are ambulatory have an EDSS of 1.0 to 4.5. Patients with impaired ambulation have scores of 5.0 to 9.5. In a review of the MS severity scores of over 6,000 patients in New York State, there has been a positive change since DMAs were introduced regardless of length of time since diagnosis.7 Kister et al confirmed this by utilizing an analysis of the EDSS distributions, adjusted for disease duration, in successive years of enrollment. The authors concluded that DMAs and better diagnosis and treatment of MS patients over the past decade have contributed to this improvement in overall scores compared to those of earlier recorded patients.7
Treatment
There is no cure for MS at the present time. Goals of therapy include decreasing symptoms, prevention or attenuation of lesion progression, and reducing long-term disabilities. Treatment includes disease-modifying therapies (DMTs) such as corticosteroids and immune modulators. Treatment of RRMS is more successful than that of the progressive forms. Since newer data suggest that the disease prognosis can be determined within the first 5 years after diagnosis, perhaps more aggressive treatment for those at highest risk should be prescribed at earlier stages of the disease.8
Current Therapies: Acute symptomatic exacerbations of MS are treated with high-dose injectable corticosteroids, usually IV methylprednisolone or dexamethasone, followed by an oral steroid taper in some cases. Response from this therapy is usually seen within 5 days of initiation.1
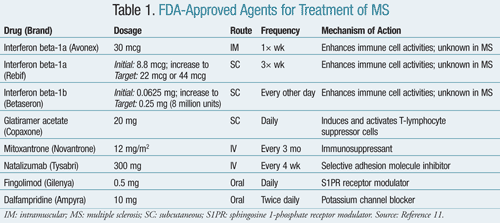
Interferon beta is considered first-line therapy for RRMS and the other forms of MS. Interferon beta-1a is available in an intramuscular (IM) and a subcutaneous (SC) formulation, and interferon beta-1b is available in a SC formulation (TABLE 1). The exact mechanism of action of interferon beta in MS is still unknown but may be related to suppression of T-helper cell response. It is thought that reducing T-cell migration into the CNS may decrease lesion proliferation. In published trials, interferon beta delayed the time to sustained progression in disability, reduced the frequency of exacerbations, and decreased subclinical disease activity as measured by MRI when compared with placebo.9,10
Interferons have significant side effects. Injection site reactions are common, as are flulike symptoms. The flulike symptoms may be treated with OTC nonsteroidal anti-inflammatory drugs (NSAIDs) and may diminish over time. Asymptomatic liver dysfunction is associated with interferon use and should be monitored at 1, 3, and 6 months and then periodically while on therapy.11
Glatiramer acetate is a mixture of four amino acids that mimics the basic myelin protein found in the myelin sheaths of nerves. It may induce T-helper type 2 (Th2)suppressor cells. No laboratory monitoring is needed. Live vaccines should not be given while on active glatiramer therapy.11 Efficacy of glatiramer is similar to that of the interferons, and this agent may be appropriate for patients with intolerance to the interferon side effects.8
Other products approved for treatment of MS include mitoxantrone and natalizumab (TABLE 1). These agents are efficacious but should be reserved for patients with progression of symptoms with prior treatment with interferons or glatiramer due to increased side effects.8 Mitoxantrone has a black box warning regarding potentially fatal heart failure and acute myelogenous leukemia (AML).11 Natalizumab has a risk of developing multifocal leukoencephalopathy, an irreversible virus-mediated demyelination syndrome that is potentially fatal.8 It must be dispensed under a risk evaluation and mitigation strategy (REMS) program with a reporting mechanism to the FDA.11
Patients are more likely to comply with an oral agent having an uncomplicated regimen than with trying to give themselves injections up to several times per week. In June 2011, the FDA approved fingolimod, the first oral DMT for MS, with monitoring using a REMS. Fingolimod may reduce the migration of lymphocytes into the CNS, which may be how it modifies the disease. Bradycardia may occur particularly following the first dose. Take a baseline heart rate and blood pressure, then monitor patients for 6 hours after the first dose to watch for signs of bradycardia and signs of atrioventricular block. If the patient becomes symptomatic, continue monitoring until back to baseline. Because of its reduction of peripheral lymphocytes to 20% to 30% of baseline, fingolimod increases the risk of life-threatening infections, especially viral illness. One month prior to initiation of therapy with fingolimod, varicella zoster virus vaccination should be given. Avoid the use of live attenuated vaccines during and for 2 months following treatment with fingolimod. There are no data supporting the use of this product in patients with diabetes. Macular edema is also a potential side effect, so an initial and periodic ophthalmologic exam is prudent.11
Walking difficulties are most often regarded as debilitating by patients with MS. Dalfampridine is an orally administered potassium channel blocker indicated to improve walking in patients with MS as demonstrated by an increase in walking speed. Walking speed was faster in feet per second over a 25-foot measured space in the dalfampridine treated patients as compared to the placebo group. Dalfampridine is contraindicated in patients with moderate or severe renal impairment and in those patients who have had a history of seizures. Seizures may occur at higher than the recommended dose (10 mg twice daily). The most common adverse events were urinary tract infections, insomnia, dizziness, headache, and nausea.12
Potential Therapies: There are several oral immunomodulator products in phase II and III studies for multiple sclerosis (TABLE 2); several of these, or similar agents, are FDA approved for a diverse group of other conditions, such as cancer, psoriasis, and rheumatoid arthritis. Cladri-bine is a purine analogue that kills some lymphocytes, preferentially CD4 cells. In the Cladribine Tablets Treating Multiple Sclerosis Orally (CLARITY) study, Cook et al observed patients with RRMS over 96 weeks who were given two short courses of drug or placebo. There were increases in infections and isolated malignancy cases in treated patients.13,14 The FDA has denied the indication for MS at this time pending further studies.14
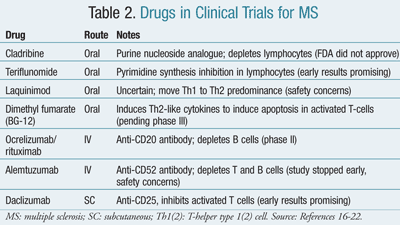
Laquinimod is an orally administered immunomodulator compound in phase III studies for MS nearing completion.15 One phase III study, Benefit-Risk Assessment of Avonex and Laquinimod (BRAVO), compared laquinimod 0.6 mg/day with interferon beta-1a 30 mcg IM every week on T2 lesion development in over 1,300 patients.16 Laquinimod reduced disability progression and fatigue over the study period. Safety parameters of the medication were similar to those of the comparator. Increased liver enzymes occur with this product. Laquinimod has been granted fast-track review status by the FDA.15,16
Teriflunomide blocks pyrimidine synthesis and reduces T- and B-cell activation, proliferation, and function in response to autoantigens. In a study of 1,000 patients over 2 years, teriflunomide reduced relapse rate, and in a higher dose (14 mg) reduced risk of disability progression. MRI-enhanced lesions were reduced on the study drug at either dose (7 or 14 mg).17 Other studies are being conducted to compare teriflunomide to interferon and efficacy after first relapse.8
Dimethyl fumarate, also known as BG-12, was compared to glatiramer in the Comparator and an Oral Fumarate in RRMS (CONFIRM) trial, which enrolled over 1,400 patients. Results showed decreased lesion formation and relapse rate. BG-12 is given 2 to 3 times a day. Most common side effects were flushing and gastrointestinal symptoms.18
Other injectable therapies now being studied in MS are also emerging. Two humanized monoclonal antibodies—alemtuzumab and daclizumab—are on the market for leukemia and kidney transplant rejection, respectively.19,20 Rituximab and ocrelizumab are chimeric monoclonal antibodies approved for lymphoma or arthritis.21,22 All of these drugs are in phase III studies, and although early findings are positive for efficacy, their side-effect profiles, including systemic immune responses and infusion reactions, may limit their use to MS patients refractory to other DMAs.8
Discussion
MS is a complex disease that requires further study to determine causative factors and new treatments that are able to better mitigate its progression. Current research is focused on finding and defining effective and safe oral medications that are able to modify the disease, as orally administered drugs also improve compliance. The available products are expanding with increasing new data and FDA approval; however, these treatments are costly and not always convenient. Each patient will have to determine, based upon the available efficacy and safety data and advice from health care providers, how they will proceed to approach this disease. Pharmacists should keep up with the latest findings to better advise patients with MS.
REFERENCES
1. Federman DD, Nabel EG, eds. Chapter 11. Neurology. Section IX. Demyelinating diseases. Multiple sclerosis. In: ACP Medicine. Philadelphia, PA: Decker Publishing Inc; 2011. STAT!Ref Online Electronic Medical Library. http://online.statref.com/
2. Alonso A, Hernán MA. Temporal trends in the incidence of multiple sclerosis: a systematic review. Neurology. 2008;71:129-135.
3. Polman CH, Reingold SC, Edan G, et al. Diagnostic criteria for multiple sclerosis: 2005 revisions to the “McDonald Criteria.” Ann Neurol. 2005;58:840-846.
4. Montalban X, Tintoré M, Swanton J, et al. MRI criteria for MS in patients with clinically isolated syndromes. Neurology. 2010;74:427-434.
5. Langer-Gould A, Popat RA, Huang SM, et al. Clinical and demographic predictors of long-term disability in patients with relapsing-remitting multiple sclerosis: a systemic review. Arch Neurol. 2006;63:1686-1691.
6. Fisniku LK, Brex PA, Altmann DR, et al. Disability and T2 MRI lesions: a 20-year follow-up of patients with relapse onset of multiple sclerosis. Brain. 2008;131:808-817.
7. Kister I, Chamot E, Bacon J, et al. Trend for decreasing Multiple Sclerosis Severity Scores (MSSS) with increasing calendar year of enrollment into the New York State Multiple Sclerosis Consortium. Mult Scler. 2011;17:725-733.
8. Gawronski KM, Rainka MM, Patel MJ, Gengo FM. Treatment options for multiple sclerosis: current and emerging therapies. Pharmacotherapy. 2010;30:916-927.
9. Kappos L, Freedman MS, Polman CH, et al. Long-term effect of early treatment with interferon beta-1b after a first clinical event suggestive of multiple sclerosis: 5-year active treatment extension of the phase 3 BENEFIT trial. Lancet Neurol. 2009;8:987-997.
10. Minagara A, Murray TJ; PROOF Study Investigators. Efficacy and tolerability of intramuscular interferon beta-1a compared with subcutaneous interferon beta-1a in relapsing MS: results from PROOF. Curr Med Res Opin. 2008;24:1049-1055.
11. Lacy CF, Armstrong LL, Goldman MP, Lance LL. Drug Information Handbook. 20th ed. Hudson, OH: Lexi-Comp, Inc; 2011.
12. FDA approves Ampyra to improve walking in adults with multiple sclerosis. FDA news release. January 22, 2010. www.fda.gov/NewsEvents/
13. Cook S, Vermersch P, Comi G, et al. Safety and tolerability of cladribine tablets in multiple sclerosis: the CLARITY study. Mult Scler. 2010;17:578-593.
14. Merck Serono receives complete response letter from FDA on cladribine tablets new drug application. March 2, 2011. www.merckserono.com/corp.
15. Jeffery S. Laquinimod Phase 3 trial meets primary endpoint in MS. Medscape Medical News. December 9, 2010. www.medscape.com/viewarticle/
16. Jeffrey S. Life still for laquinimod in MS after BRAVO. Presented at: 5th Joint Triennial Congress of the European and Americas Committees for Treatment and Research in Multiple Sclerosis (ECTRIMS/ACTRIMS); October 22, 2011; Amsterdam, Netherlands. Abstracts 148, P708, P363. Medscape Medical News. October 24, 2001. www.medscape.com/viewarticle/
17. O’Connor P, Wolinsky JS, Confavreux C. Randomized trial of oral teriflunomide for relapsing multiple sclerosis. N Engl J Med. 2011;365:1293-1303.
18. Jeffrey S. Second BG-12 trial positive in MS. Medscape Medical News. October 26, 2011. www.medscape.com/viewarticle/
19. CAMMS223 Trial Investigators. Alemtuzumab vs. interferon beta-1a in early multiple sclerosis. N Engl J Med. 2008;359:1786-1801.
20. Bielekova B, Howard T, Packer AN, et al. Effect of Anti-CD-25 antibody daclizumab in the inhibition of inflammation and stabilization of disease progression in multiple sclerosis. Arch Neurol. 2009;66:483-489.
21. Bartok B, Silverman GJ. Development of anti-CD20 therapy for multiple sclerosis. Exp Cell Res. 2011;317:1312-1318.
22. Jeffrey S. Ocrelizumab benefits in MS maintained to 96 weeks. Presented at: 5th Joint Triennial Congress of the ECTRIMS/ACTRIMS; October 20, 2011; Amsterdam, Netherlands. Abstract P462. Medscape Medical News. October 21, 2001. www.medscape.com/viewarticle/
To comment on this article, contact rdavidson@uspharmacist.com.





