US Pharm. 2015;40(2):HS18-HS24.
ABSTRACT: Nonalcoholic fatty liver disease (NAFLD) is the most common liver disorder in the United States. Current treatment options are limited; however, proper understanding of the disease and utilization of various therapeutic options are essential for management. These include nonpharmacologic and pharmacologic treatments such as lifestyle modifications, vitamin E, thiazolidinediones, and metformin. The pharmacist can play a vital role in identifying proper candidates for pharmacologic intervention and assisting with weight control, both of which are critical for management of this particular disease state.
The liver is responsible for many different physiological functions within the human body, including regulation of bile production and hematologic and metabolic systems. Within the realm of metabolic systems, one very important function of the liver is lipid metabolism. The liver regulates levels of cholesterol, triglycerides, and fatty acids within the blood.1 When dietary fats are transported to the liver, they are retained for storage. As these levels decline, the liver begins to break down reserves and releases these products into the blood. The fats are beta-oxidized to free fatty acids (FFAs) and converted to triglycerides and lipoproteins for secretion. However, defects in fatty acid metabolism can cause accumulation of fat droplets within hepatocytes and progressively abnormal lipid retention, resulting in fatty liver disease.
Fatty liver disease is the most common liver disorder in the United States, and approximately 20% of Americans have this condition without any symptoms.2 It has also been associated with complications such as type 2 diabetes, cardiovascular disease, and hepatocellular carcinoma.3 Fatty liver disease can present in one of two ways, either as a result of chronic alcohol abuse or due to years of uncontrolled hyperlipidemia, more commonly referred to as nonalcoholic fatty liver disease (NAFLD). In chronic alcoholics, levels of lipids are increased as alcohol induces lipolysis, resulting in increased transportation to the liver, and impaired lipoprotein secretion, which can progress to alcoholic fatty liver disease.4
The histopathology of NAFLD closely resembles alcoholic fatty liver disease. The pathogenesis of NAFLD, though not completely understood, involves hepatic steatosis with nonalcoholic steatohepatitis (NASH) or without hepatitis. This can eventually develop into liver cirrhosis via progression of hepatic steatosis to steatohepatitis and ultimately fibrosis, resulting in cirrhosis and liver failure.3
Unlike the pathogenesis of alcoholic fatty liver disease, NAFLD does not involve heavy or chronic alcohol consumption but instead involves triglyceride retention induced by multiple factors. These factors include reduced hepatic export of FFAs, increased import of FFA to the liver via adipose tissue, or from excessive beta-oxidation of FFA. Additional risk factors such as insulin sensitivity and metabolic syndrome also play a role.5,6
SCREENING
NASH is most often present in the setting of metabolic syndrome. Risk factors include central obesity, hyperlipidemia, hypertension, and diabetes. Patients with these conditions are at significant risk for this more severe degree of fatty inflammation and cirrhosis. The American Association for the Study of Liver Diseases (AASLD) recommends against routine screening of adults attending primary care clinics due to uncertainty of diagnostic tests and treatment options. It is also not recommended that family members of patients with fatty liver disease be routinely screened for NAFLD.7
In the event of incident findings of fatty liver, a biopsy in the absence of defined risk is not recommended. For example, if a patient has normal serum liver biochemistries and no evidence of cirrhosis but fatty liver was revealed on CT performed for another purpose, a biopsy should not be performed. These patients should be followed and monitored with laboratory testing.7
The recommendation is to consider liver biopsy when looking for definitive evidence of NASH, and, in particular, to evaluate for fibrosis or progression of risk for cirrhosis in selected patients.7 Patients who have metabolic syndrome and have suggestion of necrosis and inflammation in the liver, also known as necroinflammatory disease, are at high risk and should be considered for biopsy. These patients have abnormal serum biochemistries and metabolic syndrome. Other noninvasive tests are not recommended for routine use. One commonly used test is a NAFLD fibrosis score consisting of six variables (age, body mass index [BMI], hyperglycemia, platelet count, albumin, aspartate/alanine aminotransferase [AST/ALT] ratio), which is calculated via the published formula by Angulo et al (found at http://nafldscore.com).8 A fibrosis score may be helpful in identifying NAFLD patients with a higher probability of experiencing fibrosis or cirrhosis. However, the other metabolic indices that are commercially available are not presently recommended for routine use in clinical practice as a way of excluding NAFLD or defining risk for NASH.7
MANAGEMENT
For the patient who presents with NAFLD, treatment encompasses several different therapeutic options. In these patients, multiple comorbidities often exist such as hypertension, diabetes, hyperlipidemia, and obesity. Thus, the goal is not only to treat the liver-specific disease but to manage the comorbidities as well.
Nonpharmacologic Management
Weight Loss: The AASLD recommendation is first and foremost weight loss either with diet alone or with exercise.7 This recommendation is also in line with its guidelines for other disease states. A study published in 2010 by Promrat et al demonstrated the efficacy of weight loss with a goal of 7% to 10% in treatment-arm patients with an outcome of improved liver histology in NASH.9 Included were 31 patients stratified in a 2:1 ratio who received either intensive lifestyle modification, including diet with minimum of 200 minutes per week of exercise, or simply structured education. Outcomes included a ³3-point reduction on the NAFLD activity score (NAS) in the intensive lifestyle intervention arm. The NAS is a tool used to quantify change in NAFLD by adding up scores of steatosis (0-3), hepatocellular ballooning (0-2), and lobular inflammation (0-3), and the sum is used to provide a numerical calculation of the possibility of NASH. Patients with a >7% weight-loss reduction were shown to have significant improvement in steatosis when compared to those in the control arm (−1.36 vs. −0.41; P <.001). Accordingly, the study concluded that weight reduction achieved via intensive lifestyle intervention including behavioral modification, diet, and moderate exercise had a significant impact on liver histology in NASH patients.9
A recent study related to weight loss by Al-Jiffri et al was conducted with 100 patients with type 2 diabetes, with NAFLD stratified into two treatment arms to see what degree of weight loss was necessary for moderate improvement on liver biopsy and to insulin sensitivity.10 The treatment arm included diet modification of 1,200 kcal per day and moderate exercise of intensive treadmill activity with a goal of 65% to 70% maximum heart rate for 30 minutes at three times a week for 36 sessions over a course of 3 months. The control group had no treatment intervention.10 The study found no adverse effects related to a low caloric diet, and exercise was of moderate intensity and well tolerated. The treatment arm was shown to have a significant difference in liver enzyme tests (AST and ALT levels) and BMI. The AST was shown to have a 33.81% mean reduction (45.98 +/- 6.63 standard deviation [SD] to 34.36 +/- 5.11 SD), and ALT had a 40.8% mean reduction (46.88 +/- 5.41 SD to 33.28 +/- 4.76 SD). The study concluded that an average 15% reduction in BMI was effective in improving liver condition in type 2 diabetes patients with NAFLD.10
Weight-Loss Counseling: The pharmacist can play an important role by helping patients set weight-loss goals, which can often mean the difference between success and failure in managing NAFLD. Patients should be encouraged to set SMART goals: Specific, Measurable, Attainable, Relevant, and Time-limited. An example would be aiming to walk for 30 minutes a day, 5 days a week for the next month, and logging the results. Realistic, well-planned, weight-loss goals help keep patients focused and motivated.11
Alcohol: Along with weight loss, alcohol consumption should be reduced. The AASLD defines significant alcohol intake as more than 21 drinks per week for men and 14 drinks per week for women.7 The guidelines recommend that alcohol intake be reduced in patients who drink heavily. In patients who consume moderate amounts of alcohol, there is no definitive recommendation for reduction of alcohol intake. An occasional cocktail can be considered acceptable, as the committee stated that there was no evidence to suggest otherwise.7
Pharmacologic Management
Vitamin E: The antioxidant vitamin E has been studied, since oxidative damage has been proposed to be a key mechanism for hepatocellular injury. A meta-analysis done by Musso et al has shown conflicting data for use of vitamin E therapy.12 Of the eight randomized controlled trials (RCTs) examined, vitamin E was shown to have no benefit on liver enzymes. In addition to this, the five high-quality RCTs that were pooled were shown to have no histologic improvement. Two randomized studies in the pool did show benefit of vitamin E above placebo in terms of histologic steatosis, inflammation, and reversal of NASH in 25% of patients. These studies varied in patient population, drug dose, and treatment duration. Due to the extremely heterogeneous nature of the trials, the authors stated that it was difficult to have a firm conclusion on the effect of antioxidant therapy in NAFLD or NASH.12
A 2010 study by Sanyal et al (the PIVENS trial), consisted of 247 patients stratified across three treatment arms comparing daily vitamin E 800 IU, pioglitazone 30 mg, and placebo over 96 weeks.13 Each group had a similar number of patients distributed evenly, with comparable baseline characteristics. Primary outcome of the study was an improvement in histologic findings based on a decreased NAS score of <3, or by at least 2 points, with a 1-point decrease in either steatosis score or lobular inflammation score. Results of the primary outcome showed that patients with biopsy-proven NASH had higher rates of improvement in histologic findings via posttreatment biopsy with vitamin E as compared to placebo (42% vs. 19%; P < .001). The study concluded that for the patient population with biopsy-confirmed fibrosis without diabetes, vitamin E should be considered a first-line pharmacotherapeutic option; however, pending further evidence, it is not recommended for other patients.13
The AASLD recommends vitamin E at a daily dose of 800 IU/day as first-line therapy in nondiabetic adults with biopsy-proven NASH, as it has been shown to improve liver histology in this population.7
Thiazolidinediones: In this drug class, an option for select patients is pioglitazone. A short (6-month) study by Belfort et al specifically included 55 prediabetic or type 2 diabetes patients with confirmed NASH via liver biopsy in a randomized, double-blind, placebo-controlled trial.14 In this study, patients had similar baseline characteristics, and the treatment arm included diet plus placebo compared with diet plus pioglitazone 30 mg daily, which was increased to 45 mg after 2 months of treatment. The results included reduction in steatosis, inflammation, and necrosis of the liver in the pioglitazone treatment arm, and patients were also shown to have a 34% insulin concentration decrease and 17% fatty acid decrease compared to those on placebo. However, the known adverse effect of weight gain with thiazolidinediones was exhibited in the pioglitazone arm, and one patient presented with mild edema and discontinued therapy at 9 weeks of treatment. The authors concluded that pioglitazone had a significant impact on histologic improvement; however, it did not significantly reduce fibrosis when compared to placebo. This could have been due to the short course of the trial or because of the small number of subjects included.14
The AASLD recommends use of pioglitazone to treat steatohepatitis in patients with biopsy-proven NASH; however, long-term efficacy is not known. Pioglitazone has come under scrutiny recently due to possible association with bladder cancer when used for more than 1 year. The FDA is evaluating data from ongoing studies, but the recommendation is to avoid pioglitazone in patients with a current or personal history of bladder cancer.15 However, recent findings from a 10-year study conducted by the manufacturer demonstrate that there is no statistically significant increased risk of bladder cancer among patients ever exposed to pioglitazone.16 Consequently, a similar trial for long-term efficacy and for a better understanding of the mechanisms behind thiazolidinedione therapy is necessary to support the full application of this pharmacologic treatment.
Metformin: Another option that has been frequently reviewed is the biguanide metformin. As stated above, current pathophysiological understanding of fatty liver disease shows a correlation to insulin sensitivity.
A 12-month, open-label, randomized trial by Bugianesi et al was conducted with 110 nondiabetic patients broken into three arms, in which 55 patients received metformin 2 g per day, 28 patients received vitamin E 800 mg per day, and 27 patients received a weight-reducing diet.17 All groups were shown to have improvement in ALT associated with weight loss based on linear regression analysis. The study found that metformin patients showed decreased body weight and prompt decrease in ALT levels at 3 months. Though other controller arms showed decreased aminotransferase levels, improvement was lower or less sustained. Specifically, ALT levels at baseline in the metformin arm were 96 +/- 51 SD, and at the end of the study were reduced to 36 +/- 18 SD. The authors found that 69% of cases in the metformin group were shown to have normalized ALT, compared to 31% in the control group (P = .006). The subset of patients in the metformin and vitamin E arms showed that 44% compared to 14%, respectively, had normalized ALT (P = .019). All patients were also noted to have exceeded 90% compliance with the prescribed dose, and no patient discontinued medication because of adverse effects. The authors concluded that metformin use along with proper diet and exercise recommendations in NAFLD patients can help prevent disease progression. This study, however, was based on surrogate markers of hepatic enzyme levels and body fat, and thus the authors also mentioned that a randomized, double-blind trial would be necessary to further understand the benefit of metformin therapy in NAFLD.17
The Treatment of NAFLD in Children (TONIC) trial was a 96-week randomized, multicenter, double-dummy, placebo-controlled trial in nondiabetic pediatric patients treated with either metformin or vitamin E.18 Included were 173 pediatric patients who received either metformin 500 mg twice daily, vitamin E 400 IU twice daily, or placebo twice daily. Outcomes included serum ALT levels and histologic outcomes for steatosis based on the NAS score. The study found that both metformin and vitamin E were shown to have similar sustained ALT levels compared to placebo in the pediatric patients. ALT mean change at week 96 was −35.2 U/L (95% CI, −56.9 to −13.5) in the placebo group compared to −48.3 U/L (95% CI, −66.8 to −29.8) in the vitamin E group (P = .07) and −41.7 U/L (95% CI, −62.9 to −20.5) in the metformin group (P = .40). Though there was significant mean ALT reduction with metformin compared to placebo at week 24, the difference between the two became marginal as the study progressed. Therefore, the authors concluded that neither metformin nor vitamin E was considered to be superior to placebo in the treatment of NAFLD for the primary outcome of ALT level reduction.18
The current AASLD guidelines do not recommend metformin for treatment of NAFLD specifically.7 Treatment with metformin, however, may be beneficial in patients who have previously been diagnosed with type 2 diabetes, as it would already be in use to improve insulin sensitivity. This recommendation is based on numerous other studies that have shown conflicting data with metformin use for treatment of NAFLD. To solidify this recommendation, a randomized, double-blind, placebo-controlled trial would be necessary.7
A side note that a clinician may encounter with metformin treatment is the risk of lactic acidosis in patients with hepatic impairment.19 However, a review by Brackett clarifies this issue.20 The author states that current medical literature and prescriber information recommend caution against use of metformin in patients with liver disease. Conversely, no studies have been conducted to specifically assess the effects of metformin in patients with liver disease. All information regarding liver dysfunction is correlated via theorized mechanisms, and retrospective analyses have not shown an increased incidence of lactic acidosis. Since metformin is not intrinsically hepatotoxic, the author states that withholding the medication in patients with abnormal transaminases is not supported.20
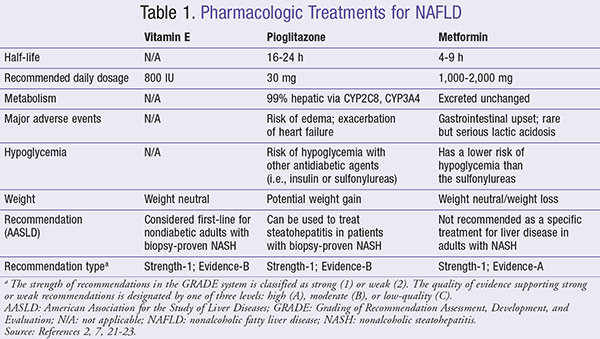
Properties of the pharmacologic treatments for NAFLD (vitamin E, pioglitazone, metformin) are summarized in TABLE 1.2,7,21-23
Potential Treatments
Omega-3 Fatty Acids: Several studies have investigated the use of omega-3 fatty acids in NAFLD for showing possible benefit; however, the results were limited by small sample size and methodological flaws. It is premature to recommend omega-3 fatty acids for the specific treatment of NAFLD or NASH, but they may be considered as the first-line agents to treat hypertriglyceridemia in patients with NAFLD.7
Statins: Several studies have established cardiovascular disease as the most common cause of death in patients with NAFLD and NASH.24 Statins are an important class of agents to treat dyslipidemia, and yet there often is resistance to using statins in patients with suspected or established chronic liver disease, including NAFLD and NASH. Given the lack of evidence to show that patients with NAFLD and NASH are at increased risk for serious drug-induced liver injury from statins, these agents should not be avoided in patients with NAFLD and NASH. Several studies have actually suggested that statins may improve liver biochemistries and histology in patients with NASH.25,26 However, there currently is no evidence to support statin use to specifically treat NASH.8
CONCLUSION
Although the pathophysiology of NAFLD is not fully understood, there is a confirmed correlation between fatty liver and other disease states such as hyperlipidemia, decreased insulin sensitivity, and obesity. Due to the lack of complete understanding, conflicting evidence makes it difficult for treatment of the disease via a pharmacologic approach. As of now, the standard of care is still weight loss via lifestyle modifications. Although more studies need to be conducted regarding treatment of NAFLD, care should be taken when prescribing certain treatments such as pioglitazone to patients because of the risk of bladder cancer and lack of literature showing long-term efficacy. Additionally, some of the efficacy studies discussed here had small populations, and most concluded that there is potential for products such as metformin and pioglitazone; however, further studies may need to be conducted before a recommendation could be made for nondiabetic patients. For patients who already have type 2 diabetes, a benefit may be seen from using a biguanide or thiazolidinedione; nevertheless, to make this recommendation, it would be ideal to see a RCT specifically involving type 2 diabetes patients with NAFLD.
REFERENCES
1. Martini F, Nath J. Fundamentals of Anatomy & Physiology. 8th ed. San Francisco, CA: Pearson Education; 2009:906-907.
2. Alldredge BK, Corelli RL, Ernst ME, et al. Applied Therapeutics: The Clinical Use of Drugs. 10th ed. Philadelphia, PA: Wolters Kluwer and Lippincott Williams & Wilkins; 2013:721-722.
3. Fruci B, Giuliano S, Mazza A, et al. Nonalcoholic fatty liver: a possible new target for type 2 diabetes prevention and treatment. Int J Mol Sci. 2013;14(11): 22933-22966.
4. Rubin R, Strayer D, Rubin E. Rubin’s Pathology: Clinicopathologic Foundations of Medicine. 5th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2008:648-649.
5. Pagano G, Pacini G, Musso G, et al. Nonalcoholic steatohepatitis, insulin resistance, and metabolic syndrome: further evidence for an etiologic association. Hepatology. 2002;35(2):367-372.
6. Marchesini G, Brizi M, Morselli-Labate AM, et al. Association of nonalcoholic fatty liver disease with insulin resistance. Am J Med. 1999;107(5):450-455.
7. Chalasani N, Younossi Z, Lavine JE, et al. The diagnosis and management of non-alcoholic fatty liver disease: practice guideline by the American Association for the Study of Liver Diseases, American College of Gastroenterology, and the American Gastroenterological Association. Hepatology. 2012;55:2005-2023.
8. Angulo P, Hui JM, Marchesini G, et al. The NAFLD fibrosis score: a noninvasive system that identifies liver fibrosis in patients with NAFLD. Hepatology. 2007;45(4): 846-854.
9. Promrat K, Kleiner D, Niemeier H, et al. Randomized controlled trial testing the effects of weight loss on nonalcoholic steatohepatitis (NASH). Hepatology. 2010;51(1):121-129.
10. Al-Jiffri O, Al-Sharif F, Abd El-Kader S, Ashmawy E. Weight reduction improves markers of hepatic function and insulin resistance in type-2 diabetic patients with non-alcoholic fatty liver. Afr Health Sci. 2013;13(3):667-672.
11. Weight loss goals: set yourself up for success. The Mayo Clinic. www.mayoclinic.org/healthy-living/weight-loss/in-depth/weight-loss/art-20048224. Accessed August 5, 2014.
12. Musso G, Gambino R, Cassader M, Pagano G. A meta-analysis of randomized trials for the treatment of nonalcoholic fatty liver disease. Hepatology. 2010;52(1): 79-104.
13. Sanyal AJ, Chalasani N, Kowdley KV, et al. Pioglitazone, vitamin E, or placebo for nonalcoholic steatohepatitis. N Engl J Med. 2010;362(18):1675-1685.
14. Belfort R, Harrison SA, Brown K, et al. A placebo-controlled trial of pioglitazone in subjects with non-alcoholic steatohepatitis. N Engl J Med. 2006;355(22): 2297-2307.
15. FDA dug safety communication: update to ongoing drug safety review of Actos (pioglitazone) and increased risk of bladder cancer. June 15, 2011. www.fda.gov/Drugs/DrugSafety/ucm259150.htm. Accessed November 7, 2014.
16. Nainggolan L. No bladder-cancer risk with pioglitazone, says Takeda. Medscape Medical News. September 4, 2014. www.medscape.com/viewarticle/831128. Accessed November 7, 2014.
17. Bugianesi E, Gentilcore E, Manini R, et al. A randomized controlled trial of metformin versus vitamin E or prescriptive diet in nonalcoholic fatty liver disease. Am J Gastroenterol. 2005;100(5):1082-1090.
18. Lavine JE, Schwimmer JB, Van Natta ML, et al. Effect of vitamin E or metformin for treatment of nonalcoholic fatty liver disease in children and adolescents: the TONIC randomized controlled trial. JAMA. 2011;305(16):1659-1668.
19. Metformin package insert. Baltimore, MD: Lupin Pharmaceuticals, Inc; July 2011.
20. Brackett CC. Clarifying metformin’s role and risks in liver dysfunction. J Am Pharm Assoc. 2010;50:407-410.
21. Katzung BG, Masters S, Trevor A. Basic and Clinical Pharmacology. 12th ed. New York, NY: McGraw-Hill Professional; 2011.
22. Brunton LB, Lazo JS, Parker KL, eds. Goodman & Gilman’s The Pharmacological Basis of Therapeutics. 11th ed. New York, NY: McGraw-Hill; 2005.
23. Lexi-Comp Online [online database]. www.lexi.com. Hudson, Ohio: Lexi-Comp, Inc. Accessed April 28, 2014.
24. Targher G, Day CP, Bonora E. Risk of cardiovascular disease in patients with nonalcoholic fatty liver disease. N Engl J Med. 2010;363:1341-1350.
25. Lewis JH, Mortensen ME, Zweig S, et al; Pravastatin in Chronic Liver Disease Study Investigators. Efficacy and safety of high-dose pravastatin in hypercholesterolemic patients with well-compensated chronic liver disease: results of a prospective, randomized, double-blind, placebo-controlled, multicenter trial. Hepatology. 2007;46:1453-1463.
26. Horlander JC, Kwo PY, Cummings OW, Koukoulis G. Atorvastatin for the treatment of NASH. Gastroenterology. 2001;120(suppl):2767.
To comment on this article, contact rdavidson@uspharmacist.com.






