US Pharm. 2015;40(12):20-23.
ABSTRACT: Probiotics are dietary supplements that have shown potential benefit in the treatment and prevention of various gastrointestinal (GI) disease states. Probiotic use may improve clinical outcomes for patients with irritable bowel syndrome, ulcerative colitis, antibiotic-associated diarrhea, hepatic encephalopathy, and portal hypertension, with minimal risk or additional adverse effects. Through the consolidation of selected scientific evidence, healthcare professionals can assist with selecting specific regimens for patients seeking to use probiotics as adjunct therapy for GI diseases.
The FDA classifies probiotics as dietary supplements, which prohibits manufacturers from making claims about efficacy in specific disease states. However, researchers continue to investigate the health benefits of probiotics and their cost-effectiveness in the treatment of gastrointestinal (GI) diseases. Considering the variety of probiotic formulations available OTC, many patients may have trouble determining the key differences between products. Due to increasing consumer interest in probiotic products, it is important for pharmacists to stay educated about the benefits of probiotics as well as which strains are most effective in each disease state according to the most recent scientific evidence.
The purpose of this article is to consolidate selected scientific evidence from randomized, controlled trials investigating the health benefits of probiotic therapy in specific GI disease states. This will allow pharmacists to help patients select the probiotic formulation most suitable for their specific GI complaints.
GI Microbiota
The term microbiota refers to a community of microorganisms populating one defined environment.1 The majority of human gut microbiota consists of members of bacterial phyla such as Bacteroidetes, Firmicutes, Proteobacteria, and Actinobacteria, with many species of anaerobic, commensal bacteria still being specifically identified.1,2 Maintenance of a healthy gut microbiota composition is thought to be important in prevention of inflammatory diseases like antibiotic-associated diarrhea (AAD), irritable bowel syndrome (IBS), and inflammatory bowel disease (IBD).3,4 These disorders theoretically derive from unnecessary activation of immune responses caused by dysbiosis, or changes in composition of resident commensal communities relative to the community of healthy individuals.1,2
Probiotics are live microorganisms that confer health benefits to the human host when adequately administered.5 They can be found in fermented foods, human milk, and manufactured supplements often available in pharmacies or online retailers.
Microbiota and probiotics are comparable in regulation and promotion of healthy immune function. Through various mechanisms, probiotics and microbiota: 1) maintain epithelial gut barrier function1,5; 2) modulate immune cell responses against pathogens1,3-6; and 3) reduce survival and virulence of pathogens.5,6 Though effects of probiotics on host microbiota
remain under investigation, reports show probiotics increase commensal microbial composition, which reduces pathogenic microorganisms.2,7 These mechanisms of action are likely responsible for the therapeutic effects of probiotic supplements in specific GI diseases as described below.
Irritable Bowel Syndrome
IBS is characterized by symptoms of chronic, episodic abdominal pain, discomfort, and altered bowel habits. The pathophysiology of IBS is not completely understood, but recent developments show symptoms may be related to genetics, diet, abnormal GI microbiota, and low-grade chronic intestinal inflammation.8
Randomized, double-blind, placebo-controlled studies evaluated probiotic effects and IBS symptomatology, including distention, bloating, flatulence, pain assessment, diarrhea, constipation, and quality of life. An international guideline from the European Society for Primary Care Gastroenterology was developed, summarizing the results to provide evidence-based recommendations.9 The guideline shows that specific probiotics may benefit patients with either diarrhea- or constipation-predominant IBS by reducing symptoms, though some studies demonstrated no positive effects. Studies with positive results recommend Bifidobacterium, Lactobacillus, Streptococcus, or Saccharomyces species in doses of 107 or 1010 colony forming units (CFU) per dose, once or twice daily, as manufactured capsules or liquids, or as fermented milk supplements.9
Ulcerative Colitis
Ulcerative colitis (UC) is a chronic inflammatory disease of the colon with symptoms of bloody diarrhea and abdominal pain. Recent scientific advances have studied genetic causes, immune and inflammatory mechanisms, and environmental factors, though the pathogenesis remains unknown. A multicentered, double-blind, randomized, placebo-controlled, parallel study tested efficacy of VSL#3 in patients with relapsing mild-to-moderate UC. VSL#3 sachets containing Lacto-bacillus paracasei, L plantarum, L acidophilus, L delbrueckii, Bifidobacterium longum, B breve, B infantis, and Streptococcus thermophilus, were added to current UC pharmacotherapy (5-amino-salicylic acid [5-ASA], azathioprine, or 6-mercaptopurine [6-MP] at fixed doses for at least 3 months before beginning the trial), and administered to patients in daily doses of 3.6 × 1011 CFU.10
Possible benefits of therapy were determined through Ulcerative Colitis Disease Activity Index (UCDAI) scores. UCDAI scores decreased 50% or more in a significantly larger percentage of patients receiving 8 weeks of VSL#3 therapy compared to placebo (63.1% vs. 40.8%; P = .031). Induction of remission occurred in 47.7% of patients administered VSL#3, compared to 32.4% of the placebo group (P = .046). Patients administered VSL#3 reported reduced rectal bleeding (P = .014); however, stool frequency did not differ between groups.10
VSL#3 treatment showed minor adverse events including dizziness, flulike symptoms, abdominal discomfort, and bloating. The placebo group reported fever, abdominal bloating, discomfort, and unpleasant taste in the mouth.10 Concomitant UC pharmacotherapy and VSL#3 administration are favored because they induce and maintain remission of UC by synergistically decreasing inflammatory mediators.11
Clostridium Difficile– and Antibiotic-Associated Diarrhea
Clostridium difficile–associated diarrhea (CDAD) and AAD are adverse events that extend a patient’s hospital stay an average of 3.6 days, costing an additional $4,000 per patient per day, significantly increasing healthcare costs and recovery time.12 A single-center, randomized, double-blind, placebo-controlled study evaluated patients receiving penicillin, cephalosporin, or clindamycin therapies and either once- or twice-daily prophylactic L acidophilus CL1285 and L casei LBC80R organisms.13 Probiotic treatment began within 36 hours of antibiotic administration and continued 5 days past the last antibiotic dose.
Compared to placebo (23.8% positive for CDAD), probiotic administration prevented CDAD in once-daily (9.4% positive for CDAD; P = .03) and twice-daily (1.2% positive for CDAD; P = .002) treatment groups. Probiotic treatment also decreased AAD symptom incidence in the twice-daily group (abdominal pain 12.8%, loose stool 31.4%, and abdominal distention 9.3%) compared to once-daily and placebo (24.7% vs. 40.5%, 44.7% vs. 58.3%, and 21.2% vs. 35.7%, respectively). Patients treated with once- or twice-daily probiotic therapy also reported shorter durations of AAD symptoms than placebo (4.1, 2.8, vs. 6.4 days, respectively; P < .05).13
Based on this and other studies with similar results, probiotics should be considered beneficial when administered to patients with concurrent broad-spectrum antibiotic use and other risk factors for AAD or CDAD.12,13
Hepatic Encephalopathy
Hepatic encephalopathy (HE) is a secondary complication of cirrhosis in which patients experience cognitive impairment and attributable mortality. Symptoms and progression of HE are associated with an imbalance of natural GI bacteria when favorable flora are replaced with harmful species.14 In response to by-products of pathogenic microbes, such as endotoxins, the immune system creates inflammatory mediators.15,16 Presence of inflammatory mediators and systemic inflammation leaves the brain more vulnerable to effects of bacterial ammonia production, and progression of HE.16
Prescription medications for HE, rifaximin and lactulose, either decrease production or increase excretion of ammonia.17 One randomized control trial compared placebo, lactulose, and a combination probiotic similar to VSL#3 containing L acidophilus, L casei, L plantarum, L delbrueckii, B longum, B breve, B infantis, and Streptococcus salivarius (thermophilus) for treatment of HE.18 After 12 months of treatment, probiotics decreased serum ammonia levels from baseline (88.2 µmol/L vs. 75.2 µmol/L; P = .04), while placebo therapy did not (89.8 µmol/L vs. 85.2 µmol/L; P = .597). Probiotics also decreased the recurrence of HE at 12-month follow-up compared to no therapy (56.9% to 34.4%; P = .001).18 There was no statistically significant difference between probiotics and lactulose therapy for decrease in ammonia levels or recurrence of HE.
Adverse effects of probiotics included abdominal distention and constipation, while lactulose side effects included diarrhea and abdominal bloating. Patients experienced probiotic side effects less frequently than lactulose therapy (36% vs. 60%).18 Given that in this study the probiotic combination showed equal efficacy as lactulose with lower risks of adverse effects, future studies can further investigate the benefits of probiotics in HE.
Reduction of Portal Hypertension
Patients with cirrhosis develop portal hypertension due to scar tissue buildup, impeding normal blood flow through the liver. Decreasing hepatic portal venous pressure is associated with fewer complications and a better prognosis. The current standard of care, beta-blocker administration, decreases portal blood pressure without fully correcting portal hypertension.19
Studies have shown norfloxacin is an appropriate adjunct therapy to propranolol to decrease portal hypertension by removing pathogenic endotoxin-producing bacteria.20 Endotoxins induce inflammatory mediators and nitric oxide, causing inflammatory and hemodynamic changes in the liver, thus increasing complications.20 However, antibiotic use can lead to resistance and additional adverse effects.
Probiotic therapy was hypothesized as an alternative by promoting growth of normal flora, which do not produce harmful endotoxins. A double-blind, randomized control trial compared propranolol plus 900 billion CFU of the probiotic VSL#3 daily to propranolol plus placebo and propranolol plus norfloxacin.19 The rate of patient response to therapy was defined as a decrease in hepatic venous pressure gradient (HVPG) >20% from baseline, or decrease to ≤12 mmHg. After 2 months of treatment, probiotic therapy increased response rates of therapy compared to placebo (60% vs. 27%; P = .025). There was no significant difference in response rates between probiotic and antibiotic treatment (60% vs. 54%; P = .776). Probiotic use decreased inflammatory mediator levels, such as tumor necrosis factor alpha (TNFα), in both peripheral (P = .005) and hepatic venous blood (P = .016) in comparison to patients that received only propranolol, with no significant difference between outcomes compared to antibiotic therapy.19
Adverse events occurred evenly across treatment groups. Upper GI bleeding occurred in one probiotic patient, two placebo patients, and one antibiotic patient. Spontaneous bacterial peritonitis (SBP) occurred in two probiotic patients, but the authors associated it with decompensated cirrhosis.19 Since probiotic use in this study showed a comparable decrease in HVPG to adjunctive norfloxacin without increasing adverse events, probiotics could be considered for adjunct therapy to decrease portal hypertension.
Risks of Probiotics
Aforementioned studies suggest probiotic side effects are mild and occur with similar frequency as the comparison treatments.10,13,18,19,21 The most common adverse effects reported in studies were abdominal distention, constipation, dizziness, flulike symptoms, abdominal discomfort and bloating, and upper GI bleeding.
Streptococcus, Bacillus, and Enterococcus are the probiotic strains with the greatest potential safety concerns because they are known human pathogens. Additionally, Lactobacillus is known to suppress T helper (Th) cell cytokine responses, affecting neonate and pregnant female immunity. L casei commonly translocates across gut membranes, which can lead to sepsis. However, probiotic-associated sepsis has only been reported in immunocompromised patients.22
Patients with pancreatitis may consider avoiding probiotics, given the results of one study showing increased mortality, though other studies did not confirm that risk.23 A recent meta-analysis on probiotics in acute pancreatitis showed no significant benefits or risks involving probiotics.24 Significant heterogeneity between studies calls for further investigation of probiotic use with pancreatitis.
Conclusion
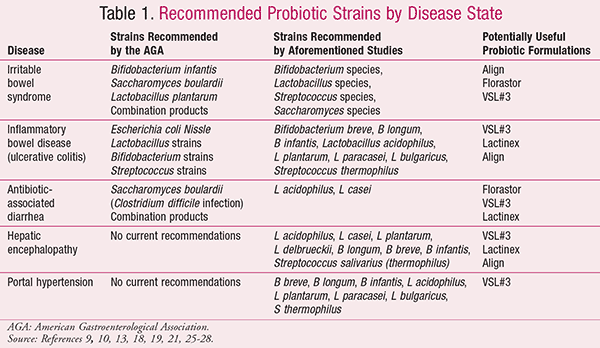
Probiotic efficacy is an expanding topic due to ease of accessibility and potential cost-efficacy for patients with varying disease states. Clinical trials of probiotics have shown potential benefits in common GI diseases such as IBS, UC, CDAD, AAD, HE, and portal hypertension (TABLE 1).9,10,13,18,19,21,25-28
Healthcare professionals can use this information to assist with the development of patient-specific recommendations. Larger scale studies are still needed to evaluate efficacy and safety in special populations, or to compare probiotic strains in head-to-head studies. Although probiotics are not currently FDA-approved as pharmacologic treatment, scientific evidence thus far indicates probiotics play an integral role in improving patient outcomes in many GI diseases.
REFERENCES
1. Thomas LV, Ockhuizen T, Suzuki K. Exploring the influence of the gut microbiota and probiotics on health: a symposium report. Br J Nutr. 2014;112(suppl 1):S1-S18.
2. Petersen C, Round JL. Defining dysbiosis and its influence on host immunity and disease. Cell Microbiol. 2014;16(7):1024-1033.
3. Plaza-Diaz J, Gomez-Llorente C, Fontana L, Gil A. Modulation of immunity and inflammatory gene expression in the gut, in inflammatory diseases of the gut and in the liver by probiotics. World J Gastroenterol. 2014;20(42):15632-15649.
4. Amalaradjou MA, Bhunia AK. Bioengineered probiotics, a strategic approach to control enteric infections. Bioengineered. 2013;4(6):379-387.
5. Ciorba MA. A gastroenterologist’s guide to probiotics. Clin Gastroenterol Hepatol. 2012;10(9):960-968.
6. Belkaid Y, Hand TW. Role of the microbiota in immunity and inflammation. Cell. 2014;157(1):121-141.
7. Fouhy F, Ross RP, Fitzgerald GF, et al. Composition of the early intestinal microbiota: knowledge, knowledge gaps and the use of high-throughput sequencing to address these gaps. Gut Microbes. 2012;3(3):203-220.
8. El-Salhy M. Recent developments in the pathophysiology of irritable bowel syndrome. World J Gastroenterol. 2015;21(25):7621-7636.
9. Hungin AP, Mulligan C, Pot B, et al. Systematic review: probiotics in the management of lower gastrointestinal symptoms in clinical practice—an evidence-based international guide. Aliment Pharmacol Ther. 2013;38(8):864-886.
10. Tursi A, Brandimarte G, Papa A, et al. Treatment of relapsing mild-to-moderate ulcerative colitis with the probiotic VSL#3 as adjunctive to a standard pharmaceutical treatment: a double-blind, randomized, placebo-controlled study. Am J Gastroenterol. 2010;105(10):2218-2227.
11. Dai C, Zheng CQ, Meng FJ, et al. VSL#3 probiotics exerts the anti-inflammatory activity via PI3k/Akt and NF-kB pathway in rat model of DSS-induced colitis. Mol Cell Biochem. 2013;374(1-2):1-11.
12. Avadhani A, Miley H. Probiotics for prevention of antibiotic-associated diarrhea and Clostridium difficile-associated disease in hospitalized adults—a meta-analysis. J Am Acad Nurse Pract. 2011;23(6):269-274.
13. Gao XW, Mubasher M, Fang CY, et al. Dose-response efficacy of a proprietary probiotic formula of Lactobacillus acidophilus CL1285 and Lactobacillus casei LBC80R for antibiotic-associated diarrhea and Clostridium difficile-associated diarrhea prophylaxis in adult patients. Am J Gastroenterol. 2010;105(7):1636-1641.
14. Kalaitzakis E. Gastrointestinal dysfunction in liver cirrhosis. World J Gastroenterol. 2014;20(40):14686-14695.
15. Bajaj JS. The role of microbiota in hepatic encephalopathy. Gut Microbes. 2014;5(3):397-403.
16. Aldridge DR, Tranah EJ, Shawcross DL. Pathogenesis of hepatic encephalopathy: role of ammonia and systemic inflammation. J Clin Exp Hepatol. 2015;5(suppl 1):S7-S20.
17. Bajaj JS. Review article: the modern management of hepatic encephalopathy. Aliment Pharmacol Ther. 2010;31(5):537-547.
18. Agrawal A, Sharma BC, Sharma P, Sarin SK. Secondary prophylaxis of hepatic encephalopathy in cirrhosis: an open-label, randomized controlled trial of lactulose, probiotics, and no therapy. Am J Gastroenterol. 2012;107(7):1043-1050.
19. Gupta N, Kumar A, Sharma P, et al. Effects of the adjunctive probiotic VSL#3 on portal haemodynamics in patients with cirrhosis and large varices: a randomized trial. Liver Int. 2013;33(8):1148-1157.
20. Fukui H. Gut-liver axis in liver cirrhosis: how to manage leaky gut and endotoxemia. World J Hepatol. 2015;7(3):425-442.
21. Probiotics: what they are and what they can do for you. American Gastroenterological Association. May 1, 2013. www.gastro.org/patient-center/brochure_Probiotics.pdf. Accessed March 5, 2015.
22. Boyle RJ, Robins-Browne RM, Tang ML. Probiotic use in clinical practice: what are the risks? Am J Clin Nutr. 2006;83(6):1256-1264.
23. Sun S, Yang K, He X, et al. Probiotics in patients with severe acute pancreatitis: a meta-analysis. Langenbeck Arch Surgery. 2009;394(1):171-177.
24. Gou S, Yang Z, Liu T, et al. Use of probiotics in the treatment of severe acute pancreatitis: a systematic review and meta-analysis of randomized controlled trials. Crit Care. 2014;18(2):R57.
25. Align information for professionals. Procter & Gamble. www.metawellness.com/en-us/hcp/align-health-care-professionals. Accessed November 5, 2015.
26. Florastor probiotics. Healthcare professionals. Biocodex. www.florastor.com/information-for-healthcare-professionals. Accessed November 5, 2015.
27. VSL#3. Scientific info. Sigma-Tau Pharmaceuticals. www.vsl3.com/hcp/scientific-info/. Accessed November 5, 2015.
28. Product center—BD Lactinex. BD (Becton, Dickinson and Company). www.bd.com/ds/productCenter/ES-Lactinex.asp. Accessed November 5, 2015.
To comment on this article, contact rdavidson@uspharmacist.com.





